Publication History
Submitted: October 08, 2023
Accepted: October 11, 2023
Published: December 11, 2023
Identification
D-0172
Citation
Sidra Arshad, Lubna Rasheed, Tasneem Kausar & Aqsa Mubeen (2023). Assessment of Cytogenotoxicity in Ctenopharyngodon Idella Injected With Endocrine Disruptors. Dinkum Journal of Natural & Scientific Innovations, 2(12):815-851.
Copyright
© 2023 DJNSI. All rights reserved
815-851
Assessment of Cytogenotoxicity in Ctenopharyngodon Idella Injected With Endocrine DisruptorsOriginal Article
Sidra Arshad 1*, Lubna Rasheed 2, Tasneem Kausar 3, Aqsa Mubeen 4
- Lahore College for Women University, Lahore, Pakistan; sidraarshad3390@gmail.com
- University of agriculture, Faisalabad, Pakistan; lubnarasheed0@gmail.com
- Lahore College for Women University, Sialkot, Pakistan; tasneemkausar0007@gmail.com
- Lahore College Women University, Lahore, Pakistan; aqsamubeen2130@gmail.com
* Correspondence: lubnarasheed0@gmail.com
Abstract: The experimental study was carried out in aquaculture facility of Department of Zoology, LCWU. Grass carp (Ctenopharyngodon idella) will be brought from Fisheries Research and Training Institute, Manawan. Fish was injected with Di-methyl phthalate for the study period of 6 days. Physiological and morphological alterations like darting, scale loss restlessness were observed in fish after the exposure to the phthalate. Growth parameters i.e. body weight and body length and their condition factor was measured and compared in fish of treated group and control group. Mean value of the condition factor for treated group was decreased as compared to the control group. At the end of experiment, blood samples was obtained to check the incidences of micronuclei and nuclear abnormalities via micronucleus assay in the peripheral erythrocytes of C. idella according to the criteria described by Fenech et al., 2003. Total of 8 types of nuclear alterations were observed in the peripheral erythrocytes of fish. Segmented nuclei were most prevalent micronuclei in blood of fish as their total percentage was 1.2% and the second most prevalent nuclei was binucleated nuclei as its total percentage was 0.9%. Clubbed shaped nuclei, micronuclei and 8 shaped nuclei had the prevalence percentage of 0.3% collectively. Blood samples obtained from control and treated group was investigated for variations in the hematological parameters. Mean concentrations of RBC’s, Hb, HCT and MCV values were elevated in the blood of the treated as compared to the control group. Statistical analysis will be performed by the application of correlation and regression analysis and ANOVA to compare the differential changes in control and treated fish groups by using SPSS version 21.
Keywords: cytogenotoxicity, ctenopharyngodon Idella, endocrine disruptors
- INTRODUCTION
Rearing of aquatic animals including crustaceans, mollusks, finfish, and seaweeds, aquaculture, the rapidly developing food producing sector in the whole world which has an approximate yearly growth rate of 5.8% under the period of 2000-2016. Production of aquaculture has been growing rapidly all over the world with an approximate “yearly rate of 8.8% since 1970. Asia presently contributes about 90% to the global production (FAO, 2018)”. It is emerging, growing and intensifying in almost every region of the world. Both the natural processes and anthropogenic activities affect the surface water quality. Freshwater contributes to the only 0.01% of the whole world water and approximately 0.8% of the surface of the Earth. Even this small portion of the water of the whole world can support minimum of 100,000 species from the 1.8 million of the 6% of all the identified species (Dudgeon et al., 2006). Streams, tributaries and rivers passing through the cities are getting large number of toxic contaminants produced from domestic/sewage, industrial and agricultural effluents (Pekey et al., 2004) which have resulted in increased degradation of fresh water ecology mainly by eutrophication. This is authentic particularly with reference to most of the evolving countries, particularly in countries of South Asia which includes Nepal Bangladesh, India, and Pakistan (Karn and Harada 2001). Industrial discharge and wastes including pesticides, heavy metals, dioxins, polychlorinated biphenyls (PCBs), petrochemicals, phenolic compounds, poly-aromatic hydrocarbons (PAHs), and microorganisms which are great hazard to our aquatic environment (Fakayode, 2005). The developing issue of litter in the aquatic environment has gained importance. It is estimated that that nearly 60–90% of marine litter is made up of plastic (Arcangeli et al., 2018).
Microplastics, which are the remnants, in diameter or length they are less than 5 mm. Microplastics are the products of plalstics introduced in the aquatic environment, so, are the confirmed form of marine contamination as they may requires centuries to be tarnished. Microplastics gather on the surface of sea, specifically in the neuston’s habitat, and one key problem is their unintentional consumption by aquatic organisms (Ryan et al., 2009). These particles after ingestion can harm an organism by affecting its chemical or physical properties and organic pollutants can also accumulate on surfaces which are hydrophobic in nature (Andersson, 2014). Its local range outspreads, from southward to northern Vietnam, coastal waters inland and southern Russia. Fish can bear water temperatures extending from 0 to 33°C, while temperatures above than 38°C has been proved lethal for adults. Grass carp are about 50-86 cm in length at maturity. Males generally have the ability to mature one year prior to females. Fins are clear to gray-brown Grass carp have a dark grey skin color on the upper surface with little lighter portions (yellow to white) and have a little golden tint (Cudmore and Mandrak, 2004).
Cytogenotoxicity biomarkers and hematological parameters (e.g., micronuclei in marginal erythrocytes and other nuclear aberrations) have been used to measure changes in fish physiology, and they have also been used for environmental biomonitoring (C¸elik et al. 2013). As there is a increased distress over the accumulation of genotoxins in the marine eviornment, the evolution of the biomarkers is quite important to recognize the genotoxins presence in the marine environment. So, it is concluded from several studies that the Comet Assay (CA) and Micronucleus (MN) test are the two rapid, sensitive and widely used processes for the recognition of mutagenicity and genotoxicity of xenobiotics and many other chemicals under laboratory and field conditions (Nwani et al., 2010). Phthalates belong to the class of universal toxicants present in the enviornment, most of these are related to the reproductive and metabolic health syndromes (Corbasson et al., 2016). In poly vinyl chloride based plastics, phthalates whenever exposed to UV radiations or weather, can conveniently detach from the plastics (Lee et al., 2006). Human biological samples like intestinal epithelial tissues, blood and pancreatic samples are identified to contain phthalates Other than that phthalates has been detected in fetal cord blood samples and in amniotic fluid, that shows phthalates can pass through placental barrier and as a result fetus is at high risk (Sathyanarayana et al., 2008). Phthalates are not easily degradable and can easily gather in various organs of a living organism. That’s why phthalates also plays as important role in the food chain by a process called as biomagnification. In biomagnification one living organism utilizes another organism as food which again utilizes another organism which is higher than the former in food chains (Chatterjee and Karlovsky, 2010).
DEHP is the most famous and widely used phthalate which is mainly used to enhance elasticity of plastics. Basically DEHP is produced from local waste water and its concentration is approximately 8-25 mgL-1. As DEHP is found in excessive amount in environment due to its high solubility and low degradation factor, the solubility of DEHP in aquatic environment is predicted to be 41 mgL-1. As 160 mgL-1 of DEHP is found in both freshwater and sea water, and this concentration of DEHP has affected the individuals adversely so only 6 mgL-1 is allowed in drinking water as highest amount. Metabolites of DEHP have the ability to bind estrogens receptors in mammals and fish therefore they exert quite harmful effects on reproduction and other capabilities of the mammals due to their reported effects. This binding property of phthalates and their compounds classify them as estrogenic chemicals. So the disrupting effects of various compounds of phthalates have been highlighted in many studies by using aquatic models. Fish has been used to assess toxological mechanisms induced by endocrine disrupting chemicals released into the environment by various elements.
In order to improvise the various properties of industrialized poly vinyl chloride like elasticity, durability, clearness and longevity, widely used phthalates can be used. (LMWPs) low-molecular-weight phthalates to (HMWPs) high-molecular-weight-phthalates the function and properties of phthalates vary accordingly. According to (Zota et al., 2014) the LMWPs having six or lower carbons such as in DEP, DMP and DBP occurs in various products of daily usage for example make up products, nail polishes and fragrances but HMWPs having more than six carbons such as DEHP, DINP, and DIDP are mainly found in the products of industrial and consumer usage for example floor tile material, pipping and glues etc (Xie et al., 2016). As in humans environment phthalates are extensively exposed (Giovanoulis et al., 2018) so human contact to phthalates is also unavoidable however they have short biological half-lives so they are detected early in humans by pseudo-steady state exposure continuously. Among many other harmful pollutants, (PAEs) esters of phthalic acid, usually called as phthalates, can be widely used in elastic PVC as plasticizer and as non-plasticizers mostly as user products, which includes food packaging, paints, medical devices, pesticides, fertilizers, and building materials (Chai et al., 2014).
On a global scale billions of tons of phthalates are generated. Studies shows that phthalates can easily migrate, leach or evaporate from the matrices in which they are used because they do not bound covalently to these plastics or any other chemical formulations used in the same process, as a result of which they can easily enter into the aquatic environment like fish, soil, foodstuff, indoor air and atmosphere (Zhang et al., 2014). In a study it was studied that phthalates have annual production of 11 billion dollars in the whole world. As phthalates can easily leach from the matrix with the passage of time and use to which they are not bounded chemically with the esters. Indoors, ambient sir, water sources and many other environmental factors, it was found that phthalates esters occur there quite frequently. Due to their presence in water sources, phthalates tend to accumulate in the fish, as fish is the main part of the food chain, as a result phthalates make their way in the food chain. Among all the other phthalates DHP has been used widely phthalate, and accounts for the 50% of the total production of phthalates. Because of its elevated increase and wide production, maximum population is suffering from hazards caused by the DEHP either by inhaling, dermal contact, ingesting, and neuro-degeneration. MEHP is the metabolized product of DEHP, it changes into the byproduct in the intestine, and this product is even more toxic than any other phthalate. The test which has been used most frequently and served as a sign cytogenetic impairment for more than 30 years is micronucleus (MN) test (Fenech et al., 2003). To check numerical and structural chromosomal abnormalities brought by aneugenic agents and clastogenic agents, MN assay is the most frequently used test (Lindberg et al., 2007).
The production of nuclear lesions (NL) reveals the abnormal ability of an organism to eliminate from the damaged nucleus, replication-failed, inappropriately condensed DNA, amplified and chromosomal fragments lacking centromeres or telomeres. Bi-nucleated cells along with nucleoplasmic bridges (BNb) are produced from di centric chromosomes, in which centromeres are pulled to the opposite poles of the isolating cell at the anaphase stage, and a nuclear membrane forms around the chromosomes (Summak et al., 2010). For detection of cytogenetic damage micronucleus (MN) assay is quite promising and accepted method. Detection of cytogenetic mutation by MN offered a very important assay to check the load in aquatic systems and pollution stress which results in the decay of populations of some specific species (Baršiene˙ et al., 2013). Nuclear damages (blebbed, lobbed, fragmenting nuclei, notched nuclei, and bi nucleated cells budding,) are measured as high quality markers for cytotoxicity. The effect of micronutrients on biota was assessed by accumulation in their tissues. However, due to chemical properties, lipophilic or amphiphilic properties, they are also biodegradable. Phthalates are mainly used as plasticizers, but also as detergents, construction products, lubricating oils, carriers for the manufacture of pesticides, solvents, personal care products, cosmetics, toys and some medical devices, soluble agents or stabilizers. Their worldwide production in 2008 was 5.2 million tonnes. Because their endocrine properties are inhibited, they have been investigated in mammals, such as mice (Kessler et al. 2004) and humans (Mortensen et al. 2005). So the present study was designed to check the injected cytotoxic impact of Di-m-phthalate to the grass carp (Ctenopharyngodon idella).
- LITERATURE REVIEW
According to Lee et al., 2019 widely used plasticizers are basically endocrine disrupting chemicals or phthalate esters which are commonly distributed in many environments. A study was designed to check the regular and longitudinal variations of atleast 14 PEs found in different environments like water, residues, air and aquatic bodies in the Lake Asan. Asan Lake is the largest synthetic Lake of Korea which is bounded by pastures and large industries. Phthalate esters were present in the whole area that is to be studied. While going downstream of the Asan Lake the amount of phthalate esters tends to be reducing in the residue samples and water. Splitting of the DEHP and DBP was calculated by means of paired sediment/water samples and fugacity fraction (ff). Large fugacity fraction value of di butyl phthalate and low fugacity fraction value of di ethyl hexyl phthalate confirmed that later one is quite abundantly found phthalate in the residue samples while former one is the second abundant phthalate other than DEHP in water. Jacobs et al., 2018 concluded that DEHP is the precursor of MEHP, which is basically a plasticizer and consistent pollutant which is also associated with oxidative stress, developing deformities and obesity.
Lee et al., 2019 demonstrated that phthalates are the commonly used endocrine disruptors and are widely accepted but not very much is known about their comparative strength. In a study approximately 6 phthalates, were selected i.e. “di-n-octyl phthalate (DnOP), dimethyl phthalate (DMP), di(2-ethylhexyl) phthalate (DEHP), diethyl phthalate (DEP), diisodecyl phthalate (DIDP), diisononyl phthalate (DINP)” and as endocrine disruptors their effects was assessed by implementing “two cell lines” and also “embryonic zebrafish assay”. Their adhesion capacity with, estrogen and the effect on, steroidogenesis was assessed with “MVLN and H295R cell assay”. In zebrafish embryos, transcription of genes regulating, steroid hormone balance, and estrogen receptors is assessed. E2/T ratio in H295R cells is significantly enhanced by introducing DnOP, DMP, DEHP, DEP. However adhesion of ERs to MLVN cells was not found. After the exposure of many phthalates, expression of “vtg1, esr1 and cyp19a1b genes in zebrafish larvae” was affected. “These results suggest that endocrine disruption in phthalate risk varies between in vitro and in vivo assays, and battery of test warrants to understand the endocrine dissociation potential of phthalates”. Aquatic organisms are widespread aesthetics, as studied by Cravedi et al., 2002. The research investigated the properties of diethylhexylphosphate (DHP) and dinofortifachalate (DBP) on trout, which are referred to as “environmental biomarkers”. Oncorhynchus mykiss (Rainbow trout) was exposed to DEHP 1mgL-1 or DBP 0.1 mgL-1 for almost 72 hours. Second group of rainbow trout was injected intraperitoneally with 50mg of DEHP or DBP on daily basis for almost 3 days. Recipient hydroxylation, ethoxyresorphin-omethylation, UDP-glucuronitransferase activity and glutathione-S-transferase activity were measured extratopathic tissues and in Liver. So it has been found that phthalate esters do not bring these and the results did not support the “induction of lauric acid hydroxide in sea bass treated with DEHP”
Phthalic acid esters (PAEs) have their lipophilic properties as studied by Xu et al., 2019. Due to the accumulation of adipose tissue in fish, a diet high in 2018 has become an important food safety issue. In a recent study, an unassuming, delicate and precise analytical procedure has been successfully made for synchronized assessment of 19 phthalic acid esters, in fish samples using “gas chromatography coupled with tandem mass spectrometry (GC-MS/MS)”. In order to determine this, a method was applied to assess 60 true fish samples obtained from the “Shanghai Municipality of China” and few phthalates like DIBP, DHP, DBP were present in all kind of fish. These sections were assessed. So the study showed that the process of marketplace monitoring of 19 PAE residues found in fish samples was appropriate. Ma et al., 2018 have shown that DHP has the ability to restrict the male reproductive endocrine system in many aquatic animals and mammals. In a recent study reproductive mechanism of male zebrafish were analyzed after exposing 3 year old male zebra fish to DEHP through their environment.
Anbumani et al., 2015 observed nuclear buds, binucleated cells, vacuolated nucleus, micronuclei, vacuolated cytoplasm and deformed nucleus under laboratory conditions for the period of 35 days in economically significant species catla catla which are basically produced by two different compounds belonging to two different classes of agrochemicals butachlor and monocrotophos in both single and combined chronic exposure under the sublethal dose of “0.625, 1.3, and 2.3 ppm and 0.016, 0.032, and 0.064 ppm”. Increase in the quantity of micronucleus and other statistically important cellular abnormalities were found. As compared to exposures of single doses, during combined exposures micronuclei frequency was doubled which demonstrated the synergistic phenomenon. When exposed to slightly lethal amount of butachlor, fish showed enucleated and binucleated cells. So it was only single study of its kind which suggested that micronuclei and other nuclear abnormalities are new likely biomarkers of cytogenotoxicity after exposures to agrochemicals suggesting their positive correlation. C. catla can be measured as appropriate aquatic bio-monitoring sentry species of polluted water bodies. Hussain et al., 2018 studied that Lebeo rohita has great economic importance due to the production of white meat, but its population in the Chenab River has been significantly reduced due to pollution. Nuclear absorption spectrophotometry showed mercury toxic levels of Mn, Pb, Cd, Sn, Zn. Cu, Zn, Hg and Cr. Comet assay results showed 42.21 31 2.06%, 31.26 41 2.41% and 21.84 21 2.21% DNA in the comet tail, 17.71 ± 1.79, 10.30 1.78 and 7.81 81 1.56 in Lebeo rohita, from three different contaminated sites on the river, 13.58 30 1.306, 8.10 and 1.04 and 5.88 6 0.06, respectively. Similar results shown by MN assay for only MN induction at 50.00 30 6.30 ay, double MN 14. 40 6 2.56 ay, while nuclear abnormalities (NA) were found to be 150.00 92–92 ay. MN stimulation and high frequency enzymes of NA were found in 96% of these fish populations in the investigational zone of the Chenab River.
Zheng et al., 2014 investigated the circulation of PAEs i.e. 15 phthalate esters in samples of sediment, deferred particles and water from Beijing’s representative lakes, and also evaluated its relationship with anthropogenic activities. Sample analysis and preparation techniques for PAE, by the help of GC-MS combined with “solid-phase extraction” or “microwave assisted extraction” and in order to check limits of detection and restorations it has been developed. Analytical outcomes of the targeted phthalate acid esters indicated that total PAE concentrations in the treated lakes extended from “0.386 to 3.184gL-1 in water”, “52.6 to 8216.4ng in dry load sediment”, and from “138.7 to 2052.4ng dry weight in suspended cells”. DEHP and DBP are mainly PAEs, which are often found in every medium, followed by DIBP and BBP. Considering their relationship to Beijing population distribution characteristics, anthropogenic activity was also found to be related with the longitudinal dispersal of phthalates in deposits. Sieriani et al., 2014 concluded that fish bios are a useful tool which can also be used to reduce the toxicity of many constituents found in aquatic environment. In fish Oryochromis niloticus selenium’s (Se) antagonistic behavior was determined against the mercury’s (Hg) toxicity. Parameters of hematology like erythroblast (ERB), Red blood cells (RBCs) and White blood cells (WBCs) in association with biomarkers of cytogenotoxicity, most commonly nuclear abberations (NA) and micronuclei (MN) after the duration of almost 3, 7, 10 and 14 days. After 7 days of exposure, cytotoxic reactions, enhanced red blood cells, leukopenia, leukocytosis and anemia were seen as the results of induction of mercury sodium selenite. Among red blood cells white blood cells erythroblasts nuclear aberrations and micronuclei affirmative correlations were found over specific period of time. So the results showed that after even exposure for a short period of time to any kind of plastic or chemical substance, variations in parameters of blood and cytogenotoxic results were observed.
Benley et al. 2016 assessed the genotoxicity was induced by less lethal amount of 10mgL-1 DBP for the time period of approximately 24 and 96 hours in Oryochromis niloticus (Nile tilapia) red blood cells. The results showed that MN frequencies are significantly (P <0.01) different in the Di Butyl “Phthalate and ethyl methane sulfonate EMS, positive control for MN bioassay groups compared to the control and solvent control groups” in the two exposure cases. The frequency of the detected nuclei was varying differently (p <0.05) when analyzing various nuclear abnormalities but the frequency of the other subtypes remained unchanged. The 96-hour exposure increased the average frequency of unidentified nuclei and the major variations between micronuclei frequencies in all groups. These results suggested that Nile tilapia have genotoxic potential at sub-lethal Di Butyl Phthalate absorptions when tested under precise research surroundings. He et al., 2020 found that phthalate esters are universally present in biotic and abiotic environmental conditions however evidence on both environmental exposure and food bioaccumulation behavior and human exposure is limited. According to a report, agricultural areas from western China were used to collect living samples to check the amount and structure of profiles of phthalates esters from indoor air, water from river and samples from open air. Di-n-butyl phthalate (DNBP) is abundant in biological specimens, in contrast to environmental models from which di- (2-ethylhexyl) phthalate is predominantly derived. A significant correlation (Pb 0.05) was detected between water of river and biota samples, showing that PAE accumulation in living sections greatly influences the river Basin. The average log of bioaccumulation factor (BAF) varies from 0.91 to 2.96, indicating that maximum PAEs are unlikely to gather in congenital creatures. The daily intake of PAEs by diet and exposure from the environment was 15, 9.4 and 1.2ngkg-1 in infants, adults and children respectively. A study by Mentor et al. 2020 suggested that exposure to endocrine disrupting chemicals could be an indication of the growing escalation trend worldwide. There are many compounds in complex chemical mixtures of human and wildlife that may have o]besogenic properties. The mixture was investigated to contain phthalate-monoesters, triclosan and perfluorinated compounds. This blend is based on serum level compounds from pregnant mother-child pregnant women in the EDC-MixRisk project. Wild type zebrafish were subjected to a 1, 10, 20, or 100-fold geometric density (HSC) mixture at five, fourteen or seventeen days post fertilization (DPF) from 3 hours post-fertilization in women. In addition, “mRNA expression of the fatty acid binding protein 11a was increased by 10 dp and 20-fold hsc mixture at 17 dpf”. One study suggests that an animal related blend of pollutants of environment disturbs the body’s metabolic rate, while “fat and lipid storage in young zebrafish provides a calorie-rich diet” hence indicating its ability to inhibit digestion. Adeogun et al., 2018 demonstrated the endocrine disruptive effects of DEHP as a waterborne source for the exposure time of almost 3, 4 and 14 days to juveniles of Clarias gariepinus by examining the patterns of transcript for estrogen receptor- α , peroxisome, aromatase and Hepatic vitellogenin (VTG) by the help of quantitative real time PCR. GC-MS technique was used to measure the load of DEHP in the liver of the fish. Tissue burden of DEHP was measured in the liver using gas chromatography-mass spectroscopy (GC-MS). Given with a sampling period of 14 days, after the histological examination of gonads, changes in gonads were evaluated in the sexes, which showed that women responded stronger than men, with no risk for DEHP at every exposed concentration. Females showed atresia of oocytes, karyoplasmic clamping ovotestis (intersex) and, while males of the fish exhibited deformity and freezing of the seminiferous tubes 14 days later in the 400 μgL-1 exposure group.
Corradetti et al., 2019 stated that “bis- (2-ethylhexyl) phthalate” (BEHP) is the most commonly used phthalate to enhance the plastic flexibility. Its metabolism has lethal effects on mammalian development and reproduction. By the help of the study, it was meant to assess the effects of 0.2-20mgL-1 of DEHP on the reproductive system of males of the zebrafish. One or three weeks later of histomorphometric examination, assessmnent of the reproductive processes and TUNEL testing, outcomes of 17b-ethynylestradiol and DEHP were determined. “DHP impaired reproduction (90%) in zebrafish by inducing mitotic arrest during spermatogenesis, increasing DNA fragmentation in sperm cells, and reducing embryo production”. Finally, low exposure to the environmental concentrations of DEHP can modify spermatogenesis and disturb zebrafish’s reproduction. Zolfaghari et al., 2014 stated that phthalates for example D (2-ethylhexyl) phthalate (DHP) are widely used as long-term plasticizers worldwide. Due to widespread use, “DEHP contains many surface waters (0.013–18.5 / g L-1), sewage (0.716–122 μg L-1), landfill leachate (88–460g L-1), sludge (12–1250 mgL-1), soil (2–10 kg)”. DEHP is dogged in the atmosphere and the toxic action of by-products caused by DEHP depletion increases the actual compound toxins. Waste water action procedures play an important part in maintaining water quality in homes, industries and rivers, lakes and reservoirs. The review addressed that the status of knowledge related to the effects of DEHP on production, incidence, fate and environment worldwide. In addition, the function and actions of Di Ethyl-hexyl phthalate in many beneficial practices including physiological, biological and advanced methods are revised and compared “qualitative and quantitative” along with the methods.
Azzouz et al., 2014 stated that “a novel analytical method using continuous solid-phase extraction system combined with gas chromatography-mass spectrometry has been reported for the simultaneous separation and determination of endocrine disruptive compounds”. This procedure has been functional to several main endocrine disrupting chemicals, including paraben, phenylphenols, alkylphenols, triclosan in water and bisphenol A. These samples were first prepared by the help of an “automated solid-phase extraction module, which consisted of a sorbent column, and analyzes with acetonitrile for derivatization with a mixture of N, O-bis (trimethyl cell) trifluoroacetamide and trimethylcalcorouzylene”. Good analytical results were found in the studied method, including low limitations (0.01–0.08 ng L-1 for 100 mL study) and good lubrication (R2> 0.997) during the concentration limits studied. This method demonstrated good accuracy (90–101% recovery) and accuracy (relative deviation less than 7%) in determining EDC in daily use of water, pond, wells, rivers, sewage and swimming pools. Large numbers and high density analyzes were found in wastewater samples (3.2–390ngL-1). As Net et al., 2015 noted that phthalic acid esters (PAEs) or phthalates are universally present in the environment because of the widespread application of these. Their occurrence has received substantial consideration due to their influence on ecology function and health of the public, that’s why their size has developed into a requirement. In order to identify these kinds of compounds, many processes of extraction and GC-MS can be used reliably. Anyhow phthalic acid esters are also found almost everywhere in research environments, which include sampling equipment, ambient air, factors, and many equipments of analytical importance, which can make it difficult to analyze actual samples with less phthalic acid esters as a background. That’s why in the environmental medium, it is a challenging task to analyze the PAEs accurately. Maximum data of literature on methods for phthalic acid esters quantification in non-artificial media has been revised in the review of a paper. Sample extraction, detection and sample were reviewed and compared to determine the amount of PAE in many other medium of the environment like air, sludge, water and soil etc.
Giovanoulis et al., 2018, explained the use of phthalates esters in many applications as plasticizers. Humans are exposed to many phthalates like di methyl phthalate, di butyl phthalate, di-n-butyl phthalate and by using an extensive data record from the studied population of Norway, their occurrence in human’s atmosphere can be detected which provides not only the significance of some other paths but also explains the average intake evaluation. Ingestion, especially inhalation, skin absorption, and oral intake were assessed by a variety of means, and the total intake was calculated from bio monitoring data based on all rapid means. The relative importance of different fast paths has been compared with different studies, where inhalation is significant for phthalates having less molecular weight and insignificant for phthalates having large molecular weight. Dermatological consumption based on dermal ingestion by dust, air and products of personal care is very low (median 2000 times). However, skin regeneration is not a highly studied risk trail and many research breaks (such as immersion fractions) continue. However, intensive care for phthalates like DPHP and DINCH is required as their consumption is most likely to increase in near future. Zhu et al., 2016 studies that PAEs are often found in marine environments. Effects on the transcriptional summaries of important genes intricate with vitellogenin (VTG), sex steroids and steroidogenesis have been examined. Outcomes show that production of eggs and quality of sperm decrease after MEHP exposure, resulting in reduction of diameter of eggs and reduced egg content also reduction of protein content of eggs. Substantial stimulation of testosterone of plasma and 17-isotole estradiol (E2) in women was observed, resulting in decreased CYP19a and 17β-HSD gene transcript at the ovary. Other than that, level of plasma vitelloglycan was pointedly enhanced after their contact with MEHP in both of the sexes. Furthermore, in F1 embryos, after persistent contact with MEHP leads to the increased rates of hatching and malformations in contrast to those embryos that have not introduced with MEHP. Results has shown that this specific phthalate i.e. MEHP has the ability to disturb the offspring’s growth and also has the ability to produce reproductive deformities. Though, very significant effects have been witnessed at greater applications, and it may not have a large effect on fish reproduction and development, usually at measured concentrations.
Zhai et al., 2014 stated that phthalates are widely used as a plastic in everyday life, which resulted in their extended circulation in many environments. Yet, there is only little data available on how phthalates has effects on endocrine disruption in aquatic organisms. The study was to investigate whether “the hydrolytic metabolite of D- (2-ethylhexyl) phthalate (DEHP) disrupts the thyroid endocrine system in fish after exposure to mono- (2-ethylhexyl) phthalate (MEHP)”. Embryos of Zebrafish (Danio rerio) were introduced with diverse amounts of MEHP (1.6, 8, 40, and 200 μg L-1) after 2 hours of fertilization to 168 hours of fertilization. “The whole body content of thyroid hormones and the transcription of genes involved in the hypothalamic – pituitary – thyroid (HPT) axis have been investigated”. Total content of the body T4 has been drastically reduced and T3 increased whole body content showed the endocrine disruption of thyroids. Overall, all the consequences of the study showed that under high introduction to MEHP to fish larvae / embryos can alter the total proportion of the body’s thyroid hormones and transcriptional changes in genes which have roles in hypothalamic pituitary thyroid, thereby eliminating the “endocrine toxicity” of thyroids.
Adeniyi et al., 2011 stated that by the use of “flame ionization gas chromatography”, esters of four phthalates can be quantified and identified i.e. DBP, DMP. DEHP, DEP in fishes, water and sediments. From Ogun river catchments, these collections were gathered. The di methyl phthalate was the only phthalate which was not detected in fish and water samples but it was only detected in sediments samples collected from six different sites of sampling. Concentrations of DEHP, DBP and DEP in fish samples ranged from “320.0–810.0, 380.0–1,080.0, and 40.0–150.0 μgkg-1 in Tilapia sp.; 310.0–860.0, 400.0–1,170.0, and 40.0–110.0 μgkg-1 in Chrysichthys sp and 320.0–810.0, 400.0–3,970.0, and 30.0–300.0 μgkg-1 (DEHP) in Synodontis sp” respectively. Accumulation of phthalate esters is entirely independent of fish species which is a signal that differences in the levels of phthalate in fish were not statistically legit at p < 0.05. In case of drinking water the amount of DEHP, DBP and DEP was relatively high as compared to the concentrations allowed by the environment protection agency of USA. Pollution index of phthalate and accumulation in the bio sediments were also evaluated.
Phthalates have an extensive use in consumer and industrial consumer products too as stated by Chen et al., 2008. Great potency as reproductive toxicants between all the phthalates is shown by DEHP and DBP. The aim of the study was to examine the level of migration of different phthalates from films of Poly Vinyl Chloride films and by putting on handling of food and phthalate burden in the body for Taiwanese. The estimation of phthalate esters movement was determined by covering the food with PVC films and then heated in microwave. As depicted by all the results of the study it was estimated that level of DEHP has been increased significantly after introducing it with heat for 3 minutes straight. In the controlled condition of heating, the considered consumption of phthalate and their “tolerable daily intake (TDI, based on body weight of 60 kg) from eating one 400-g meal were 1705.6 μg and 92.2% for DEHP”. So it was concluded from the experiment that DEHP’s burden in the body for Taiwanese in this region, reveals that materials made up of plastics has been used intensively.
In order to determine the relationship among level of phthalate in residue and marine factors and accumulation factor of bio-residues in 17 rivers of Taiwan, concentration of phthalate compounds and esters in fishes were estimated as stated by Huang et al., 2008. Average amount of DEHP, DBP and BBzP in residues at “low-flow season were 4.1 (<0.05–46.5), 0.22 (<0.05–3.1) and 0.14 (<0.05–1.3) mg kg−1 dw; those at high-flow season were 1.2 (<0.05–13.1), 0.13 (<0.05–0.27) and 0.09 (<0.05–0.22) mg kg−1 dw, respectively”. However, traces of DMP, DOP and DEP were present in both seasons in sediments. Factors which were affecting the amount of DEHP in residues were mainly suspended solids, chemical oxygen demand (COD), temperature and nitrogen-ammonia. Among two species e.g. Oreochromis miloticus niloticus and Liza subviridis highest amount of DEHP was found. In sediments of rivers, concentration of DEHP was highly affected by parameters of water quality because they have great process of bio-degradation as our results suggested and also that in fish level of DEHP was greatly influenced by contaminated pollutant’s physiochemical factors and habitat of the fish. Immunotoxic properties of DBP were studied by Xu et al., 2015 by using albino and transgenic zebrafish. Formation of immune cells and ability to phagocytize in embryos of zebrafish was analyzed after exposing them to different amounts of DBP from six hours after fertilization to 72 or 96 hpf. Formation of neutrophils and macrophages in dose-dependent method was found to be constrained. However, if immunotoxicity has occurred or not it can be inferred from weather the nacrophage’s ability to phagocytize has been decreased after the introduction to DBP or not. Trancription levels cell-related genes T/B was all increased and there was the the induction of respiratory burst also. However, results shows that in order to determine the developmental toxicity from toxic chemicals in immune system, embryos of zebrafish serves as a great tool and that di butyl phthalate in aquatic enviornment has greatly affected the fish’s immune system.
According to Fatoki et al., 2010 phthalate ester plasticizers have been suggested in South Africa’s Venda region. In order to separate them by quantitative analysis different techniques were used which were “liquid-liquid extraction, column chromatographic clean-up and capillary gas chromatography”. Different values for the amount of phthalate esters in the water and in sediments were observed. Generally DBP and DEHP had the highest concentrations in water and sediments because of their large extent of use in plastics and many other chemicals of industrial use. The optimum phthalate level in water samples is maximum 3µgt-1 but in these samples phthalate level was increased from this value recommended by environmental protection agency of USA for the safe survival of fish and other aquatic bodies. Exposure to phthalates in any case can cause serious health problems like cancer and many other toxic issues. Phthalates found in water can cause these problems as untreated water maybe consumed by the public either as absorption through skin or through freshwater. As toxic effects of DEHP has revealed by its use or exposure pathway so DEHP is known to have caused the highest risk of many diseases as compared to the other phthalates. Phthalates results in water samples causing environmental concern as public health is declining if rural people use this water.
Valton et al., 2014 for the first time investigated seven phthalate by-products, in aimed tissues of roach from a polluted river of the Ile-de-France district. Bioaccumulation of all parent phthalates was investigated in liver and river concentrations (p<0.01) were correlated with liver contents. In plasma, bile and liver, all metabolites were observerd. The mono-n-butyl phthalate (MnBP; 1.5 μgg-1 dw of liver) , lead to mono-iso-butyl phthalate (MiBP; 1.6 μgg-1 dw of liver), were the most plentiful. Among the three metabolites of diethylhexyl phthalate (DEHP), mono (2-ethyl5-hydroxyhexyl) phthalate – MEHHP (214 ng ml-1) is predominated in plasma while in liver and bile it was mono (2-ethylhexyl) phthalate (MEHP). The ratio of MEHP/DEHP in liver ranged from 0.04 to 0.2. Only mono (2-ethyl-5-oxohexyl) phthalate (MEOHP) was associated (p<0.05) with parent DEHP among the oxidized metabolites and as compare to monoester it seemed to be a more consistent marker of DEHP effect. Cravedi and Durand, 2002 studied the wide exposure of aquatic organisms to phthalate esters. Impacts of dibutylphthalate (DBP) and diethylhexylphthalate (DEHP) are studied in trout on potential environmental bioindicators , xenobiotic metabolizing enzymes. Waterborne exposure of DBP (0.1 or 1 mg/l) or DEHP (1 mg/l) for 72 hrs to Rainbow trout (Oncorhynchus mykiss) was done. An intraperitoneal insertion of 50 mg kg-1 of DEHP or DBP was given to a second group of rainbow trout daily for three days. Different activities like Ethoxyresorufin-odeethylation, Laurate hydroxylation, glutathione-S-transferase and UDP-glucuronyl transferase were calculated in extrahepatic tissues and liver. Specially, the phthalate esters are considered as not the stimulaters of these enzymes, the formerly interpreted results of sea bass induced with lauric acid hydroxylase treated with DEHP does not support it. Santamaria et al., 2016 evaluated that in recent decades the effect of environmental contamination on aquatic organisms is alarming. Genotoxicity and Haematological biomarkers were assessed in two local fish species’ (Astyanax gr. bimaculatus and Aequidens metae) peripheral blood. The study was conducted at three various sites of Ocoa River during dry and rainy season. The sites are called Nacimiento(site 1), Centauros (site 2), Caño Seco (site 3). Sampling of fish from a reference location having slight possibility of infectivity was also done. Variations like reduction in erythrocyte number, lymphocytes, haemoglobin amount, increased neutrophils and thrombocytes counts, along with nuclear anomalies in erythrocytes like high micronuclei number , notched, blebbed and lobed nuclei and binucleate cells were found in fish that was taken from site 3, pursued by the site 2 in rainy season. The outcomes exhibited that genotoxic and haematological changes occurred in the monitored organisms of sites 2 and 3 which obtained Indus-trial and household wastewater during rainy season.
Seriani et al., 2015 concluded that the using biomarkers for fish’s biomonitoring shows a valuable means for the evaluation of aquatic contamination. Sampling of Water and fish (Oreochromis niloticus) was done from a control site and from a pond in the Parque Ecológico do Tietê (PET) that is located along the Tietê River (São Paulo, Brazil). Fish were utilized to observe the characteristics of blood and gill mucus. Considerably increased counts of erythrocytes (RBC), erythroblasts, leukocytes (WBC), lymphocytes and Mean Corpuscular Volume (MCV) were shown by hematological parameter analyzes of PET fish as compared to the controls; though, notably lesser Mean Corpuscular Hemoglobin Concentration (MCHC) values and hemoglobin content was observed. The number of micronuclei and nuclear anomalies were considerably higher. This is recommended by results that fish taken from the polluted site reveal a sequence of physiological responses, which apparently express health problems.
Barsiene et al., 2013 determined the evaluation of ecological genotoxicity and cytotoxicity levels in the framework of the ICON project in blood erythrocytes of haddock (Melanogrammus aeglefinus) and dab (Limanda limanda) which obtained in August–October 2008 near the coast of Iceland and at 25 stations in the North Sea. Environmental genotoxicity bioindicators like nuclear buds, micronuclei and bi-nucleated cells with nucleoplasmic bridges were evaluated, and bi-nucleated and fragmented-apoptotic erythrocytes’ occurrence were determined as ecological cytotoxicity bioindicators. Frequencies of cytotoxicity and genotoxicity responses were considerably lesser in fish from the Icelandic reference area as compare to North Sea areas fish. Barkaa et al., 2016 applied micronucleus (MN) test for the evaluation of genotoxicity in microcrustaceans. Copepods (Acanthocyclops robustus) and Daphnids (Daphnia magna) were obtained in situ and acclimatized for 24 hrs in the lab. The micronucleus didn’t work for Copepods but it was successful for Daphnids. Exposure of sublethal amounts of insecticide (deltamethrin) or metals (Cu, Zn, Cd) was given to mature Daphnids for 2 and 7 days. After 2 days of exposure, dose-dependent induction of MN was examined, induction at the maximum doses increased by 2-fold for every chemical tested. In genotoxicity evaluation use of daphnids is highlighted because of its advantages and ecological significance. For aquatic genotoxicity hazard estimation, the Daphnid test is a reliable assay as well as a helpful choice for amphibian studies.
Mankidya et al., 2013 concluded that chemical factors like phthalic acid esters are utilized to improve the plasticity of industrial polymers. Endocrine disruption cytotoxicity, lipid peroxidation, effects mediated through AhR and effects on enzyme’s expression of xenobiotic metabolism induced by benzyl butyl phthalate (BBP), di-(2-ethy hexyl) phthalate (DEHP), dibutyl phthalate (DBP) and diethyl phthalate (DEP) were evaluated in growing fish embryos. In transactivation studies estradiol (E2) is not mimiced by DEP and DEHP. Weak effectiveness was revealed by all phthalates as agonists of the aryl hydrocarbon receptor (AhR). Four phthalates potency order was; DEHP > DEP > BBP » DBP. This study highlighted the requirement for immediate evaluation of: (1) effect of phthalates on numerous cellular targets and (2) when several phthalates adapt similar pathway, phthalate mixtures considered to exhibit additive effects. Anbumani and Mohankumar, 2015 studied cytogenotoxic effects in the economically valuable fish Catla catla of India. The Cytogenotoxic effects like deformed nucleus and micronuclei, binucleated cells, nuclear buds, vacuolated cytoplasm, vacuolated nucleus, enucleus and echinocytes were activated by two compounds which belong to two different chemical classes of agrochemicals (monocrotophos and butachlor) at sublethal quantities (0.625, 1.3, and 2.3 ppm and 0.016, 0.032, and 0.064 ppm). The chronic exposure was separate as well as joined under laboratory settings for the duration of 35 days. The results showed cytological abnormalities and critical development in the numbers of micronucleus (MN) which was duration dependent. Enucleated and Binucleated cells were observed just in those fishes which exposed to sublethal quantities of butachlor. The study recommended that in polluted water bodies, Catla catla is measured as an appropriate aquatic biomonitoring specie.
Ahmed and Thompson, 2019 concluded that in order to contribute food security and human diet, fast growth of aquaculture has been examined as a huge change, that is an access to growing production of fish universally. The blue water consumption in aquaculture is also considered to be involved in worldwide production of fish. Although, a broad range of ecological concerns, including water pollution, eutrophication, habitat devastation, ecological effects, disease onset and biotic reduction are thought to be linked with the huge revolution of aquaculture. Additionally, because of shrimp cultivation, blue carbon (i.e., carbon in coastal and marine ecosystems) discharges from mangrove deforestation are gathering. Aquaculture need to develop continuously in order to grow production of fish for increasing universal population along with its ecological effects should decrease prominently. Cheng et al., 2019 investigated 13 phthalate esters’(PAEs) parallel profiles, quantities and geographical distribution in the aquatic fish ponds of Pearl River Delta (PRD) site. Sampling of water and sediments was done from 22 areas in the duration of July 2016 to september 2017. In sediment and water samples, the major compound was di-2-ethylehexyle phthalate (DEHP). It accounted for 70.1% and 66.1% of PPAEs in water and sediment, respectively. The quantities of SPAEs in sediment of estuary and river in the PRD region (p < 0.05) was lower than in sediment. The median amounts of di-n-butyl phthalate (DBP) and DEHP increased which suggest environmental risk limit (ERL) which exhibited a possible danger to the environment of aquaculture fish pond in the PRD.
Malem et al., 2019 investigated potential pollution caused by phthalates in sediments and seawater. The study was conducted in the region of Rayong, Chanthaburi and Chonburi which are situated near the eastern coast of Thailand. Benzyl butyl phthalate (BBP) and diethyl phthalate (DEP) among other six chosen phthalates were not observed in a single sample, whereas the phthalates that observed in negligible or very low concentrations in most samples were di-n-octyle and dimethyl phthalates. In December 2014, diethylhexyl (DEHP) and dibutyl phthalate (DBP) quantities were 0.23–0.77 and 0.31–0.91 μgL-1 in seawater, respectively around the region of Pradu Bay. In sediment samples, 11 out of 20 were non-detected (ND)-0.80 and ND-1.65 μgg-1. Significant congeners that measured were DEHP and DBP. Hussain et al., 2018 determined that Labeo rohita is commercially very significant due to production of white meat, however because of increasing pollution in Chenab River the population of this fish has been decreased considerably. 40 ± 2.56% were shown by micronucleus test, whereas nuclear anomalies were observed as 150.00 ± 2.92%. The reason of 96% decrease of this fish species population in the experimental region of River Chenab was the greatest number of nuclear abnormalities and induction of MN. Along the River Chenab, this fish species is observed close to extinction. Labeo rohita exhibited high responsiveness to pollution and may be used as a biomarker due to its large scale extinction. Benli et al., 2016 assessed genotoxicity after disclosure to little amount of di-n-butyl phthalate about1 0mgL-1 for the durations of 24 and 96 hours in the erythrocytes of Nile tilapia (Oreochromis niloticus). In both schemes of exposure, it was exhibited by results that mean micronuclei numbers in ethyl methane-sulfonate and DBP groups were predominantly different (p<0.01) regarding solvent control and control groups. The numbers of other subtypes were not modified but the number of damaged nuclei was considerably different (p<0.05) during the evaluation of nuclear anomalies. Induction of prominent variations among MN numbers in all groups (p<0.01) and expansion in mean numbers of notched nuclei was observed after exposure of 96 hours. Results indicated that when analyzed under regulated conditions of laboratory, sub lethal quantities of DBP have genotoxic possibilities for Nile tilapia.
Li et al., 2015 stated that red tide algae have been shown to inhibit immune action and damage mechanisms of dibutyl phthalate (DBP) on Karenia brevis. Results showed that algae experienced oxidative stress after being exposed to 5 mg L P 1 DBP. Malondioaldehyde (MDA) peaked after 72 h, about 2.3 times higher for untreated cells. Superoxide dismutase (SOD) and catalyst (cat) activity increased significantly after 48 hours as a positive response. DBP induced high production of reactive oxygen species (ROS), with a maximum H2O2 content of 250 nmol/107 cells at a maximum of 33 U mL, 48 h per 1 hour, and 72 Nm as OH; The next two values are 2.5 and 4.4 times higher than those observed for the controls, respectively. TEM images showed that many small vacuoles or apical tubers were commonly seen around the cell membrane and that the membrane structure was eventually broken down. Further experiments were performed to identify the original ROS production sites after DBP exposure. Controls of CuZn-SOD (mainly cytosolic isoforms, also found in some chloroplasts) are 2.5 times higher than controls under DBP exposure, although Mn-SOD (mitochondrial isoform) activity is significantly inhibited. No significant difference was observed in the activity of Fe-SOD (chloroplastic isoform). In addition, dicumarol (inhibitor of the electron transport chain across the plasma membrane) stimulated DBP-induced ROS production, while rotenone (inhibitor of mitochondrial electron transport chain complex I) reduced DBP-induced ROS production. These results suggest that mitochondria may be the main target sites for DBP attack.
In polyvinyl chloride industry Di-(2-ethylhexyl) phthalate (DEHP) is used as a plasticizer in the whole world as studied by Wang et al., 2018b. Existence of DEHP in all the aquatic and marine environments has recorded for years. In aquatic organisms, toxicological effects of DEHP have still not been researched thoroughly. In embryos of fully frown up Daphnia magna which has been exposed to DEHP, acute toxicity, antioxidant activities of enzymes, expression patterns of genes of antioxidant enzymes were investigated. It was found that the “median lethal concentrations (LC50) of DEHP for juveniles exposed for 24 and 48 h were 0.83 and 0.56 mg L−1, respectively”. The Lethal concentration of DEHP introduced in adults for 1 and 2 days were 0.48 and 0.35 mg L−1. When compared to control group, Daphnia magna which was exposed to DEHP showed an increased monoaldehyde levels for 24 h and also lower average antioxidant capacity. During prolong exposures and high doses, enzyme activity was nearly reduced, but on 24 h earlier exposure, levels of antioxidants enzymes and glutathione S-transferases was increased. So the results showed that organic and functional effects has been seen in both adults and juvenile D. magna, and changes in the both levels of transcription in genes and peroxidation of lipids has increased. Singh et al., 2011 determined that phthalates were widely used as plasticizers to increase the softness and flexibility of polyvinyl chloride plastics, although they could penetrate the surrounding environment. There is considerable evidence in rats that phthalate exposure causes developmental and reproductive toxicity. Curated interactions between 16 phthalates and genes / proteins were obtained from the Comparative Toxicogenomics Database (CTD), and a total of 445 curated phthalates (DHP / MEPP and DBP / BBP / MBP) and 249 specific genes / proteins were found to interact. In genetics, the pathways and networks of these 249 unique genes / proteins are thoroughly analyzed. The pathways and networks of the top 34 genes / proteins were found to be similar to those of 249 specific genes / proteins. Therefore, the top 34 genes / proteins act as molecular biomarkers of phthalate toxicity. The first three phthalate toxicity categories are identified as cardiotoxicity, hepatotoxicity and nephrotoxicity and the top 20 diseases are cardiac, liver, urological, endocrine and genital diseases.
Ye et al., 2014 predicted that apprehensions have been raised about the adverse effects of di- (2-ethylhexyl) -phthalate (DEHP) on reproduction. However, limited information is available on the effects of DEHP on marine organisms. The aim of the present study is to investigate whether prolonged exposure to DEHP and its active metabolite mono- (2-ethylhexyl) -phthalate (MEHP) may impair endocrine function in marine medica (Origias melestigma). Sea urchin larvae are exposed to DEHP (0.1 and 0.5 mg / L) or MEHP (0.1 and 0.5 mg / L) for 6 months and have effects on reproduction, sex steroid hormones, liver vitellogenin (VTG), gonad histology and Gone. The expression of genes involved in the hypothalamic-pituitary-gonad (HPG) axis has been investigated. Exposure to DEHP, but not MEHP, accelerated the onset of spawning from hatching to puberty and decreased egg production in exposed females. Furthermore, exposure to both DEHP and MEHP reduced the rate of fertilization of untreated female ovarian cysts paired with untreated males. Therefore, DEHP has a more pronounced toxic effect compared to MEHP. These results suggest that exposure to DEHP and MEHP from puberty to adolescence may lead to endocrine disruption with a sex-specific effect on sea urchins, in which women outnumber men.
Li et al., 2014 stated that D- (2-ethylhexyl) -phthalate (DHP) is universal uses endocrine disruptor. It has widespread contact in the general population, which has caused considerable public concern due to its detrimental health effects. . It is appropriate to investigate the molecular mechanisms of its testicular toxicity, which are largely unknown. By feeding DEHP to male rats for 2 weeks, rat spermatogenesis was impaired, resulting in decreased sperm count and sperm count. We investigated the effect of DEHP on DNA replication because rapidly dividing tissue is particularly susceptible to DEHP toxicity. Surprisingly, DEHP inhibits DNA replication as revealed by fiber tract analysis results. This led to the stimulation of mitochondrial apoptotic pathways and increased ROS production. Furthermore, the toxicity of DEHP may be due to respiratory chain defects and attenuation of ATP levels brought about by hyperparalysis and underarm SIRT1 activity. Our research reveals previously unknown mitochondrial dysfunction in DEHP-induced testicular toxicity and highlights the importance of SIRT1 in male reproduction.
Three ensemble groups of juvenile yellow catfish were subjected to three levels of DEHP (0, 0.1 and 0.5 mg L− 1) for 56 days as studied by Yuan et al., 2017 for Pelatobagrus fulvidraco. Fish survival (100%) was not affected by different levels of ambient DEHP. Fish feed intake is high for final body weight, weight gain, specific growth rate and exposure to 0.5 mgL-1 DEHP. In contrast, the hepatosomatic index of fish was 0.1 and 0.5 mgL-1 were the lowest exposed to DEHP. Serum total protein, glutamic – pyruvic transaminase, glutamic – oxaloacetic transaminase, glucose and triglycerides increased with increasing concentrations of DEHP exposure. Fish exposed to 0.5 mg L− 1 DEHP had the lowest superoxide dismutase and glutathione peroxidase activity, while fish malondioaldehyde content was higher than fish exposed to 0.1 and 0.5 mg L− 1 DEHP. The phagocytic indices of the control group were highest. After intravenous injection with Aeromonas hydrophila, fish in the control group had the highest expression of toll, such as receptor 5, and the lowest expression of 88 myeloid differentiation factors in fish exposed to 0.5 mg L-DH. The study suggests that it may interfere with hepatic metabolism, induce ROS production, and lead to the reduction of blood toxicity and immunity by the accumulation of malondialdehyde.
Zanotelli et al., 2009 stated that Di 2 (ethylhexyl) phthalate (DEHP) is a extensively used plasticizer that is generally found to be polluting the aquatic environment. Hence, much less is known about the long-term effects of DEHP on fish development. Therefore, we aim to investigate the effects of DHEP (i) on densities in the environment and (ii) conditions that cause more lethal consequences (larvae) than adult fish (using higher temperatures). Different concentrations of DEHP (0.1–10 µg l−1rpar) applied continuously for 91 days were tested on guppies less than one week of age at the beginning of treatment. 14 days after the exposure began, Guppy was treated. DEHP with 10 10g l DE 1 showed a shorter body length than the control fish. Its effect on body weight was more pronounced when it was reduced by 40 and 70% at 1 and 10 −g l DE DEHP, respectively. The reduction in growth was still significant at 91 days of DEHP treatment, although the Fulton condition factor was not affected, while DEHP inhibited the growth of male and female infections, with no change in sexual development. . These statistics are in DEHP. Concentrations in the aquatic environment can severely affect fish growth.
Dong et al., 2018 stated that bisphenol A (BPA) and Di-N-butyl phthalate (DBP), endocrine-disrupting chemicals (EDCS) are harmful to human health. Frequent exposure to BPA and DBP causes human disease. Despite the potential risk to fetal development, the mechanism of action of BPA and DBP on vertebrate development and disease is still unclear. In the present study, we identified proteins and protein networks that are affected by BPA and DBP during zebrafish (Danio rerio) development. Zebrafish embryos were exposed to environmental levels of BPA (10 μgL-1) and DBP (50 gL-1) for 96 hours. Quantitative proteomics labeling by ITRAQ identified a set of 26 and 41 different proteins in zebrafish embryos treated with BPA- and DBP-, respectively. Integrated Toxicity Analysis predicts that these proteins will function in general control networks that are significantly associated with developmental and metabolic disorders. Exposure to low concentrations of BPA and DBP poses health risks to zebrafish embryos. Our results suggest that BPA and DBP significantly regulate the expression levels of many network proteins, providing valuable information about the molecular functions of BPA and DBP on developmental systems. Gardener et al., 2017 stated that the phthalates and compounds used to add plastic elasticity are ubiquitous in the environment. In particular, diethyl (DEP), D-N-propyl (DN-NPP), and D-N-butyl (DBP) phthalates have been found to obtain harmful effects in mammalian and non-mammalian studies, including toxicity. The effects vary according to the alkyl chain length. The embryo of the African claw frog Xenopus lavis has been used to assess the toxicity and teratogenicity of many compounds and serves as a model for estimating the adverse and teratogenic effects of ortho-phthalate esters. The purpose of this study was to develop a model for the comparison of the toxic effects of the development of ortho-phthalate esters using genotype embryos. This study highlighted the development of Xenopus lewis embryos with concentrations of DEP, DNPP, and DBP using 96-h malignant concentrations and deformities of 96-h frog embryo teratogenesis assay-xenopus (FETXA). And concentrations that inhibit the increase indicated that DEP, DNPP and DBP showed improved toxicity with increasing Ester length. The development of Xenopus lewis exposed to DEP, DNPP and DBP exhibits similar distortions, which increase the alkali chain length at low concentrations. The teratogenic risk did not change significantly with the alkyl chain length, with data showing that only DBP was teratogenic. Qu et al., 2015 performed laboratory experiments have been performed to determine antioxidant responses to nine phthalates (PAEs) in the liver of goldfish Carassius aratus. Fish were injected with 1 mg of each PAE and 10 mg / kg body weight on days 4, 8 and 15. The potential biotoxicity of PAE was investigated using antioxidant and lipid peroxide indicators. We found that the levels of superoxide dismutase (SOD), catalyst (CAT), glutathione peroxidase (GPx) and malondihydride (MDA) exhibited different trends after prolonged treatment, indicating that the metabolism was less toxic and produces more active ingredients. Based on the enzyme inhibition, MDA content, and severity of the calculated integrated biomarker reaction (IBR), the toxic sequence was determined as follows: di-butyl phthalate (DBP)> diethyl phthalate (DIDP)> diphenyl phthalate. In particular, DBP, which exhibits significant inhibition of enzyme activity and a great decrease in MDA content, may be highly toxic contaminants. Furthermore, our results suggest that IBR may be a common marker of contamination.
Huang et al., 2016 stated that Di-n-butyl phthalate (DBP) and D-2-ethylhexyl phthalate (DEHP) are the two most commonly used phthalates, while Cu (II) is the normal equilibrium state of copper. They are found everywhere in the aquatic environment, but very little is known about their mixed toxicity to aquatic organisms. In this study, we assessed the combined effects of these two phthalates of copper and Daphnia magna and photobacterium phosphorium, the most severe toxicity expressed by EC50 (concentration causes 50% of the maximum effect). Toxicity sequence was DEHP + Cu (II)> DBP + Cu (II)> Cu (II)> DEHP> DBP for both test strains. Adverse effects have been found in the mixed toxicity of Qi (II) with DBP or DEHP using the toxicity unit method. These researches have significant implications in the environmental risk assessment for phthalates in the aquatic environment in the presence of heavy metals. Khalil et al., 2016, noted that the use of consumer goods such as plasticizers has significantly increased the presence of genetic antibiotics such as thalic acid esters (PE) in freshwater and marine environments due to the regular release of contaminated waterlogging and waste. Hepato-renal markers [alanine aminotransferase activity (ALT), creatinine and urea levels] and cortisol levels were also determined in serum. In addition, the liver, kidneys, and gill tissues were examined histopathologically. Comparative results between the 1/2 96-h LC50 group and the 1/3 96-h LC50 group clearly showed a significant increase in MDA levels and a significant increase in antioxidant barriers as well as DNA damage. Elevated hepatocellular markers and cortisol levels have also been observed. Histopathological examination of the liver, kidneys, and gills showed pathological changes associated with changes in the biochemical profile of the exposed fish. In addition, asymmetric clinical signs have been identified. Based on these results, we conclude from our study that exposure of larvae O. niloticus to DBP may induce biochemical and tissue morphological changes leading to oxidative injury and DNA damage.
Sayom et al., 2018 claims that phthalates are being used as plasticizers to increase the durability, resistance and flexibility of plastic materials. DEHP has been linked to various toxins in humans and animals, resulting in the formation of other phthalates such as dibutyl phthalate (DBP) and diethyl phthalate (DEP). The increasing use of phthalates has polluted the aquatic ecosystem and is a direct threat to aquatic life. In this study, we analyzed the effects of three phthalates DEHP, DEP and DBP using freshwater organism Daphnia Magna. However, lactation and incubation delays did not occur due to exposure to three phthalates at 1 and 10μM, with prolonged exposure to 14 days reducing body length. As indicated by Oil Red O Staining, fat accumulation is increased by exposure to all phthalates. QRT-PCR analysis of genes involved in fat metabolism suggests that the increase in fat content may be due to inhibition of fatty acid absorption and catabolism. Fertility analysis revealed that DBP and DEP did not change honesty, but surprisingly, reproduction increased 1.5-fold compared to the DEHP control group at 1μM. Significantly reduced the negative effect of 10μM on phthalates lifetime and both 1 and 10μM lifetime on DBP. In addition to limited phthalate DEHP, our data indicate that other alternative phthalates DEP and DBP may also have harmful effects on aquatic organisms.
Lu et al., 2017 explored the effect of DBP on early embryonic production in Sicily Marine invertebrates, Galleria caspitosa. DBP was detected by a symptomatic pattern of abnormalities to induce sperm dysfunction and induce weak and defective embryogenesis. Thus, after the first rupture, one of these abnormal embryos was able to carry the blastomere further into the mitoses, while the second was arrested. Analysis of microtubules, chromosomes, and actin filaments reveals that in abnormal embryos the mitotic spindles are irregularly bent, small, and unable to anchor to the cortex, resulting in faulty isolation of chromosomes. In undifferentiated blastomeres, karyokinosis has been found to persist slowly, suggesting the presence of multiple sets of mitotic spindles. However, cytokinesis in these arrested cells is impaired, failing to collect the contractile actin ring, resulting in one pole of the embryo forming a larger, more distinct cell. DBP was found to suppress the action of superoxide dismutase in spermatozoa, and with this modification, DBP-treated cells experienced oxidative stress, as indicated by the presence of lipid aldehyde in the sperm acrosome, such as 4-hydroxinonanol (4) -HNE. And neck. This research emphasizes the oxidative damage of spermatozoa and discovers novel potential mechanisms for regenerative poisoning by lipid aldehydes, including alkalis of epithelial structures in spermatozoa.
- MATERIALS AND METHODS
The present study was conducted in the Aquaculture Facility, Department of Zoology, Lahore College for Women University, Lahore.
3.1. PHASE I: Collection, acclimatization of fish and experimental set up
Healthy juveniles of Grass carp (Ctenopharyngodon idella) (n=60) approx. 150-200 g in weight and 4-5 inches in length, were collected from Fisheries Research and Training Institute, Manawan, Lahore and then transferred to the aquaculture facility at Department of Zoology, LCWU. Fish was kept under facility conditions in order to get acclimatized to the new environment for one week (Figure 3.1). Exposed fish were randomly sampled at the durations of 3 days. 1/4th water was changed from each aquarium on daily basis in order to remove any fecal contamination. Fish blood samples was obtained and kept in EDTA tubes for further investigations.
3.1.1. EXPERIMENTAL SET UP
Fish sample was divided into two main groups i.e. treated group and control group to study the phthalates exposure.
- Control group (TO)
Fish of the control group were not exposed to any kind of phthalates and were given normal diet @3% of fish body weight (Figure 3.2).
- Treated group (T)
Treated group was divided into further two groups T1 and T2 (Figure 3.3). The fish of the treated group were injected intramuscularly with the 1.5µgL-1 concentration of Di-methyl phthalate (Figure 3.4) for 6 days.
- Preparation of Dilute Di-Methyl Phthalate
Di-methyl phthalate is basically an organic compound with the chemical formula of (C₂H₃O₂)₂C₆H₄. It is a colorless liquid and soluble in organic solvents. In order to prepare the required dose of di-methyl phthalate 1.5ml of di-methyl phthalate was diluted with 98ml of distilled water under room temperature (Figure 3.4).

Figure 3.1: Experimental set up

Figure 3.2: Control group

Figure 3.3: Treated group

Figure 3.4: Preparation of Di-Methyl phthalate
3.2. PHASE II: PHYSIOLOGICAL ALTERATIONS IN FISH AFTER EXPOSURE
After injecting the fish with Di-methyl phthalate, fish was observed to record any kind of physiologoical changes that occurs in the fish after the exposure. Morphological and physiological changes that may occur in the fish were recorded in the phase III. After exposing fish to the phthalate for 6 days, following parameters will be observed during the trial period; mucus secretion, pigmentation, scale loss, lack of balance, darting, swimming pattern, and mortality.
3.3 PHASE III: WEIGHT-LENGTH RELATIONSHIP AND CONDITION FACTOR
Total health of the fish will be assessed after the exposure to the Di-methyl-phthalate for the 6 days. 5 fish samples were selected after the trial period to analyze the growth parameters like body length and body weight and their condition factor will be calculated. Figure 3.5 and 3.4 shows the measurement of the body length and body weight of the fish during the trial period.

Figure 3.5: Body length measurements

Figure 3.6: Body weight measurements
3.3. PHASE IV: HAEMATOLOGICAL ANALYSIS
At the end of experimental trial, the blood samples, obtained from control fish and treated fish was transferred to Falcon tubes and investigated for variations in the hematological parameters i.e. White Blood Cells, Red Blood Cells, Mean corpuscle volume MCV, Mean Cell hemoglobin MCH, by following the formulae of Shah and Altindag, 2004.
- Total RBC count
By using an improved Neubaur haemocytometer total amount of red blood cells were counted. Erythrocytes were counted by loading in the haemocytometer and their total number was reported.
- Total WBC count
Total number of white blood cells was determined by using an improved Neubaur haemocytometer.
- Estimation of Hemoglobin
By the help of hemoglobin test kit (DIAGNOVA), hemoglobin (Hb) was determined by Cyanmethemoglobin method.
- Estimation of Packed Cell Volume (PCV) % value
Packed cell volume (PCV) % was calculated by Wintrobes tube method.
- Estimation of Mean Corpuscular Volume (MCV)
This is the average volume of red blood cells. As the size of the cell is very small it is written in cubic microns (µm3). MCV was calculated by using the formula:
Mean Corpuscular Volume MCV (µm3) =
- Estimation of Mean Corpuscular Hemoglobin Concentration MCHC
Mean Corpuscular Hemoglobin Concentration is basically the measure of the average concentration of Hemoglobin in a given volume of packed red blood cell. MCHC can be calculated by the formula:
Mean Corpuscular hemoglobin Concentration MCHC (gdL-1) =
- Estimation of Mean Corpuscular Volume MCH
The mean corpuscular volume, or mean cell volume, is a measure of the average volume of a red blood corpuscle. The measure is attained by multiplying a volume of blood by the proportion of blood that is cellular, and dividing that product by the number of erythrocytes in that volume. MCH can be calculated by the formula:
MCH (pg) =
3.4. PHASE V: MICRONUCLEUS ANALYSIS
Cytogenotoxicity analyzing was done through MN assay which is an easy genotoxic study technique. Nuclear lesions and micro nuclei formed by phthalates in fish blood cells were observed under fluorescent microscope. For that purpose a little amount of blood from fish was taken and a thick blood smear was made on glass slide to detect the presence of micronuclei in multiple cells by following the methods of fenech et al., 2003.
3.4.1. Peripheral Blood Sample Extraction
In order to analyze the micronuclei formed as a result of introduction of fish to phthalates, fish will be anaesthetized by clove oil. EDTA is used as a coagulant for syringes in order to avoid any coagulation. Blood samples from both control and treated groups were taken by the help of syringes from caudal vein (Figure 3.7). Collected blood was instantly dropped on labeled slides in order to make thick smear (Figure 3.8)
3.4.2. Fixation of Slides
Coverslips are used to cover the smeared slides for 4-5 minutes and then allowed to dry. After drying, fixation was done by fixing slides in methanol for about 10 minutes. Slides were taken out carefully from the fixation cassettes and leave them air dried for 2-3 minutes (Figure 3.9).
3.4.3. Staining of the Slides
After the process of fixation, staining of slides was performed, in which slides were stained in 10% Giemsa with phosphate buffer for about 15 minutes in cassettes (Figure 3.10). After staining slides were rinsed with distilled water and air dried after rinsing at room temperature. After staining slides were observed under fluorescent microscopes in order to observe and count micronuclei and nuclear lesions.

Figure 3.7: Collection of blood samples
Figure 3.8: Smear on slide

Figure 3.9: Fixation of slides

Figure 3.10: Staining of slides
3.4.4. Nuclear Lesions
Nuclear lesions were analyzed for the determination of the prevalence percentage and frequency in the di methyl treated group. Nuclear lesions were divided into seven major types with the following criteria: a) Lobed nuclei (LN) containing distinct lobes, b) Notched nuclei (NN) with some depth into nucleus, c) Bi-nucleated (BN) consisting of two nuclei, d) Multinucleated, e) Clubbed shaped nuclei, f) Smaller circular or oval bodies of chromatin were considered as micronuclei, and g) Eight shaped nuclei.
The MN frequency was calculated by the following formula:
MN%= ×100
3.4.5. Induction of Micronuclei
Small extranuclear bodies which are the results of whole chromosomes which were unable to reach spindles during cell division and chromosome breaks leads to the formation of micronuclei (MN). According to Volders et al., 2002, at telophase, these chromosomes fragments which were unable to reach spindles, they do not combine with the main nucleus but instead compressed into another small micronuclei. Mutagens can be the main cause of MN induction, but it can induce impulsively. In normal progenitor cells, the main example of impulsively formed MN is chromosome loss which than results in induction of MN. Large variety of mechanisms epigenetic or genetic both can induce MN. Any exposure to clastogens or aneugens can result in the formation of micronuclei or they can also be produced spontaneously as a result to cell differentiation.
3.4.6. Survival and Fate of Micronuclei
Mateuca et al. 2006 has described the possible outcomes of the MN after its formation. Postmitotic fate includes: as extra-nuclear activity MN may retain in the cytoplasm of the cell, when it has finished one or two rounds of chromosome/DNA replication, MN may incorporate the main nucleus and it might restart usual biological activities, and as a result of apoptosis MN may disappear or eliminate from the cell.
3.5. PHASE VI: STATISTICAL ANALYSIS
The statistical analysis of the data will be performed after experiments. The SPSS version 21 will be used for the application of linear regression and correlation and analysis of variance (ANOVA). All the results will be represented by mean values and standard deviations in order to identify the variations between means of the values of all the treated groups.
- RESULTS
Grass carp (Ctenopharyngodon idella) was collected from Fisheries Research and Training Institute, Manawan, Lahore and then injected with di methyl phthalate to study the endocrine disruption potential of the introduced phthalate. Micronucleus assay and hematological analysis was performed to check the abnormalities in the peripheral blood cells of the fish.
4.1. PHYSICAL CHANGES IN FISH AFTER EXPOSURE
No adverse physical changes or mortality was observed in the fish after exposure to 1.5 µgL-1 concentration of Di-methyl phthalate for 6 days. No pigmentation or changing of color on either side of the fish was observed. There was no mucus secretion. Scale loss was observed during the trial period. However micro changes occurred in the behavior of the fish which are as follows: lack of balance, agitated or erratic swimming, air gulping, restlessness and jumping, and sudden quick movement.
4.2. WEIGHT-LENGTH RELATIONSHIP AND CONDITION FACTOR
To analyze the fish health status and pattern of growth before and after the exposure, length weight relationship and condition factors were evaluate. In order to determine the weight-length relationship, multiple fish samples were selected which showed the variable length and weight values. Examined specimens exhibited total length and weight varying between 13.5-11.4cm (Mean=13.98±1.69) 24-14.5g (Mean=24.1±6.45). Minimum body weight and length recorded in treated group was 14.5g and 11.4cm respectively. Similarly, minimum body weight and length recorded in control group was 20g and 12cm respectively. While maximum body weight and length in treated group was 30g and 15.5cm. Similarly in control group highest body weight and length was 39g and 158cm respectively. Values of both sample size and length and weight are presented in Table 4.1 and 4.2.
Table 4.1: Values of body weight (g), body length (cm) and condition factor (CF gcm-3) of the fish (Ctenopharyngodon idella) injected with di methyl phthalate
| Body Weight
(g) |
Body length
(cm) |
Condition factor
K=WL-3 ×100 |
| 24 | 13.5 | 0.97 |
| 30 | 15.5 | 0.81 |
| 22 | 14 | 0.80 |
| 30 | 15.5 | 0.81 |
| 14.5 | 11.4 | 0.98 |
| Mean= 24.1±6.45 | Mean= 13.98±1.69 | Mean= 0.88±0.09 |
Table 4.2: Values of body weight (g), body length (cm) and condition factor (CF gcm-3) of the control group of fish (Ctenopharyngodon idella)
| Body Weight
(g) |
Body length
(cm) |
Condition factor
K=WL-3 ×100 |
| 39 | 15.8 | 0.98 |
| 20 | 14.5 | 0.65 |
| 24 | 12.8 | 1.14 |
| 25 | 12 | 1.44 |
| 29 | 13 | 1.31 |
| Mean= 34±7.23 | Mean= 14.4±1.97 | Mean= 1.15±0.23 |
Length-weight relationship of the fish is used to calculate the growth patterns of fish while condition factor is used to verify the health conditions of the fish after exposure to di methyl phthalate. Length weight values as well as condition factor were used to evaluate the growth pattern and health status of fish injected with endocrine disruptors. Condition factor of Ctenopharyngodon idella was calculated by the formula K= (WL-3) ×100. Values of condition factor for both treated and control group was calculated. Values varied from 0.80gcm-3 to 0.98gcm-3. While values of condition factor for control group was varied from 0.98gcm-3 to 1.31gcm-3. The highest value for condition factor in treated was for the fish having weight and length was 14.5g d 11.4cm. Similarly in control group fish having body weight and length of 25g and cm respectively showed the highest growth factor of 1.44gcm-3. Mean value for the condition factor in treated group was 0.88±0.09 gcm-3 and mean value for condition factor in control group was 1.15±0.23 gcm-3 respectively. As condition factor is the overall health parameter of a fish so overall condition of the fish having the least value for condition factor in treated group with weight and length of 22g and 14cm was 0.81 and this showed the poor health of the fish. In control group however, the condition factor for the fish having weight and length of 25g and 12cm respectively showed the highest condition factor of 1.44gcm-3. Figure 4.1 shows different parameters of growth i.e. body weight and body length of the fish injected with di methyl phthalate.
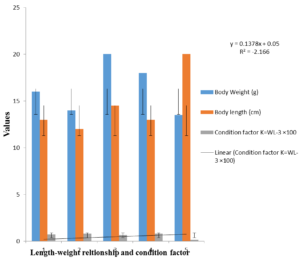
Figure 4.1: Comparison of biometric data and health condition of fish Ctenopharyngodon idella after injected with di methyl phthalate
4.3. HEMATOLOGICAL PARAMETERS
After exposure of the fish to the di methyl phthalate for 6 days blood from the control and treated groups was taken and analyzed for the detection of the hematological parameters like RBC’s, HGB, HCT, PLT, MCV, MCH, MCHC. Blood was taken from caudal region of the fish and transferred to EDTA tubes. These samples taken from the treated and control group were used to estimate the hematological parameters. MCV, MCH and MCHC were counted by using the values of RBC’s, HB and HCT by following the formulae given in (Shah and Altindag, 2004).
4.3.1 Total RBC count
Total RBC count was made by using hemocytometer by using the method of (Shah and Altindag, 2004). Erythrocytes were counted in loaded hemocytometer chamber and total mean value was 0.07±0.028. While in control group total erythrocyte count was 0.04×106/µL which is less than treated group. Elevation in RBC’s level in treated group can be a consequence of body’s stress response to the toxicant to which fish was exposed. Due to the elevation in the RBC’s level other hematological parameters like HB and HCT were also increased
4.3.2 Hemoglobin Content
Control group of the fish showed the total value of 2.1g/dL while the fish from the treated group showed the mean value of 4.0g/dL. Treated group showed a significant increase in the hemoglobin level due to the increase in the RBC’s level. As treated group showed increased level of hemoglobin value it is because of the increased value of RBC’s in the treated fish.
4.3.3 Total Hematocrit count (HCT)
Mean value of the hematocrit count determined for treated group was 0.3±0.14 % while HCT count for control group was 0.2%. HCT count for treated group was elevated as compared to control group. Total HCT count was increased in the fish as fish responded to the toxicant i.e. Di-methyl-phthalate.
4.3.4 Total Mean Corpuscular Volume (MCV)
In treated group mean MCV count was 42±2.82 while in control group total MCV value was recorded as 50.0×106 µm3. In treated group mean corpuscular volume has significantly decreased as compared to control group. MCV value has decreased in the treated group because fish responded to the toxicant injected in the fish.
4.3.5 Total Mean Corpuscular Hemoglobin (MCH)
In treated group mean MCH count was 6.2±2.54 while in control group total MCH value was recorded as 5.25×108/µm3. In treated group mean corpuscular volume has slightly increased as compared to control group. Total Mean Corpuscular Hemoglobin (MCH) values has increased in treated group because of the elevation in the RBC’s value because of the stress response generated in fish after the exposure to Di-methyl-phthalate.
4.3.6 Total Mean Corpuscular Hemoglobin (MCHC)
In treated group mean MCHC count was 1500±707.1 while in control group total MCHC value was recorded as 1050gdL-1. In treated group mean corpuscular volume has significantly increased as compared to control group. Elevated values of MCHC in fish of treated group indicated that fish has generated an in immune response to the toxicant to which it was exposed. Elevation in the different hematological parameters showed that the fish has responded to the di-methyl-phthalate by enhancing the level of RBC’s, Hb, HCT, MCV and MCHC as compared to the control group. Value of mean hemoglobin concentration (MCH) has decreased in treated group as compared to the control group after the exposure of fish to di-methyl-phthalate. Values of different hematological parameters of control and treated group of the fish are mentioned in Table 4.3.
Table 4.3: Hematological Parameters of control and treated group of the fish (Ctenopharyngodon idella)
|
Parameters |
Fish Group | |
| Control | Treated | |
| RBC’s (×106/µL) | 0.04 | 0.07±0.028 |
| HGB (g/dL) | 2.1 | 4.0 |
| HCT (%) | 0.2 | 0.3±0.14 |
| PLT (×103/µL) | 848 | 1227.5±225.6 |
| MCV (×106 µm3) | 50.0 | 42±2.82 |
| MCH (×108 µm3) | 5.25 | 6.2±2.54 |
| MCHC (gdL-1) | 1050 | 1500±707.1 |
4.4 Micronucleus assay for the assessment of nuclear alterations in peripheral erythrocytes of the fish (Ctenopharyngodon idella) injected with Di methyl phthalate
Fish (Ctenopharyngodon idella) that was brought from Fisheries research and training institute, Manawan, Lahore was injected with di methyl phthalate for 6 days. After exposure, blood from caudal region of the fish was taken and transferred to EDTA tubes for further observations. A thin smear of the blood was made on glass slide and then observed under fluorescent microscope. Micronucleus assay was performed to check the toxicity induced in the peripheral erythrocytes of the fish injected with di-methyl-phthalate. As a result of exposure to the toxicant, nuclear abnormalities were induced in the blood of the fish in the form of many micronuclei. Nuclear abnormalities were induced in the fish after exposure to phthalate i.e. Di-methyl-phthalate for 6 days. The most frequent and prevalent nuclear alteration were segmented and binucleated nuclei as they were highest in frequency with 0.9 and 1.2% of micronuclei as compared to the other micronuclear lesions examined in the thin smears of the blood samples. It means that exposure to the toxicant like phthalate can induce nuclear lesions and can alter the shape of the micronuclei. Micronuclei, clubbed shaped nuclei and dumbbelled shaped nuclei were of the same mean frequency of 0.3%. Total mean frequency of the nuclear lesions was 0.62±0.32. Figure 4.2 shows the induction of Notched nuclei in the erythrocytes of the fresh water fish are produced as a result of exposure to the di-methyl-phthalate. Figure 4.3 shows the induction of Dumbelled shaped nuclei (DN) observed in the blood of freshwater fish injected with di-methyl-phthalate. Figure 4.4 showed the notched shaped nuclei produced as a result of exposure to the toxicant. Figure 4.5 showed nuclear alteration like Binucleated nuclei (BN), segmented nuclei (SN) and Micronuclei (MN) in the blood of freshwater fish injected with di-methyl-phthalate Figure 4.6 showed the nuclear alterations, Binucleated nuclei (BN), segmented nuclei (SN) Notched shaped (NN), Kidney shaped nuclei (KN) and 8 shaped nuclei (EN) observed in the blood of fish. Figure 4.7 showed the Notched nuclei (NN) and Binucleated nuclei (BN) in fish erythrocytes after exposure.
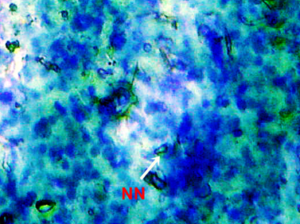
Figure 4.2: Micronucleus test and nuclear alterations, Notched nuclei (NN) observed in the blood of freshwater fish injected with di-methyl-phthalate
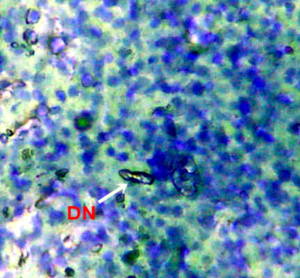
Figure 4.3: Micronucleus test and nuclear alterations, Dumbelled shaped nuclei (DN) observed in the blood of freshwater fish injected with di-methyl-phthalate
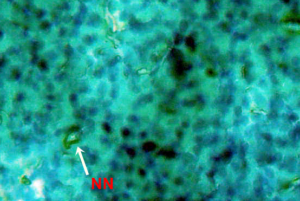
Figure 4.4: Micronucleus test and nuclear alterations, Notched nuclei (NN) observed in the blood of freshwater fish injected with di-methyl-phthalate
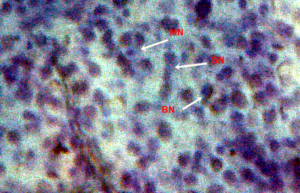
Figure 4.5: Micronucleus test and nuclear alterations, Binucleated nuclei (BN), segmented nuclei (SN) and Micronuclei (MN) observed in the blood of freshwater fish injected with di-methyl-phthalate
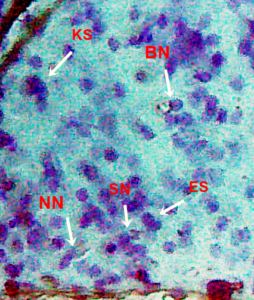
Figure 4.6: Micronucleus test and nuclear alterations, Binucleated nuclei (BN), segmented nuclei (SN) Notched shaped (NN), Kidney shaped nuclei (KN) and 8 shaped nuclei (EN) observed in the blood of freshwater fish injected with di-methyl-phthalate
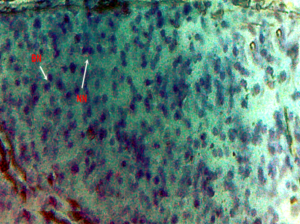
Figure 4.7: Micronucleus test and nuclear alterations, Notched nuclei (NN) and Binucleated nuclei (BN) observed in the blood of freshwater fish injected with di-methyl-phthalate
Different shapes of micronuclei and nuclear lesions were observed in the peripheral erythrocytes of the fish. Eight major nuclear alterations were observed which includes notched shaped nuclei (Figure 4.2, 4.4, 4.6 and 4.7), clubbed shaped, binucleated (Figure 4.5, 4.6, 4.7, 4.8, and 4.9), kidney shaped (Figure 4.6 and 4.8), segmented nuclei (Figure 4.5 and 4.6), micronuclei (Figure 4.5 and 4.9), 8 shaped nuclei (Figure 4.6) and dumbbell shaped (Figure 4.3) . Total 1150 cells were counted in each slide of the treated and control group that was made by the thin smears of the blood of the fish. Figure 4.8 showed the Micronucleus test and nuclear alterations like Kidney shaped nuclei (KN) and Binucleated nuclei (BN) in the blood of freshwater fish injected with di-methyl-phthalate. Figure 4.9 showed nuclear alterations like, micronuclei (MN) and Binucleated nuclei (BN) observed in the blood of freshwater fish after its exposure to di-methyl-phthalate for the period of 6 days. The least prevalent micronuclei in the fish erythrocytes were clubbed shaped nuclei and micronuclei with total percentage of MN as 0.3%. However 8 shaped micronuclei and binucleated nuclei had the total frequency of 0.7% and 0.9% respectively. 8 shaped micronuclei and binucleated nuclei had almost the same prevalent frequency. Frequency of kidney shaped micronuclei was 0.6%. By following the results it is concluded that nuclear lesions in blood cells followed the order:
MN= SN>BN>NN>EN>KN>CN>DN>MN
Frequencies of different nuclear lesions induced by di methyl phthalate in the treated group of the fish are given in table 4.4.

Figure 4.8: Micronucleus test and nuclear alterations, Kidney shaped nuclei (KN) and Binucleated nuclei (BN) observed in the blood of freshwater fish injected with di-methyl-phthalate
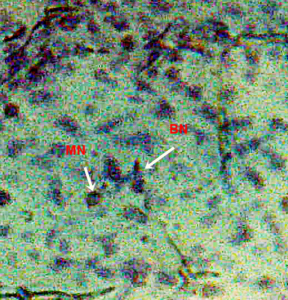
Figure 4.9: Micronucleus test and nuclear alterations, micronuclei (MN) and Binucleated nuclei (BN) observed in the blood of freshwater fish injected with di-methyl-phthalate
Table 4.4: Frequency of micronuclei induced in fish erythrocytes after the exposure
| Types of Micronuclei
(MN) |
Total number of cells
(n) |
Frequency of micronuclei
(MN) |
% of MN |
| Notched nuclei | 1150 | 9 | 0.7 |
| Clubbed nuclei | 1150 | 4 | 0.3 |
| Bi-nucleated nuclei | 1150 | 11 | 0.9 |
| Kidney shaped nuclei | 1150 | 7 | 0.6 |
| Dumbbell shaped nuclei | 1150 | 4 | 0.3 |
| 8 shaped nuclei | 1150 | 8 | 0.7 |
| Segmented nuclei | 1150 | 14 | 1.2 |
| Micronuclei | 1150 | 4 | 0.3 |
| Mean±S.D | 7.62±3.66 | 0.62±0.32 |
- DISCUSSION
Rivers, tributaries and streams are receiving a large number of contaminants and these contaminants often include plasticizers, discharge from different industries containing different contaminants and phthalates etc. These contaminants are not only affecting the environment but also impose great hazard to all the aquatic living organisms. Large number of anthropogenic chemicals entering the water on this extent is quite hazardous to aquatic organisms. Effect of different phthalates in order to check the health status of the fish was investigating by assessing cytogenotoxicity in Ctenopharyngodon idella injected with endocrine disruptors, di methyl phthalate in the fisheries and Aquaculture laboratory of Lahore College for Women University. Genotoxicity studies of chemical substances are widely used to identify the adverse effects of chemicals with respect to DNA damages in the form of gene mutation, recombination, numerical changes and structural chromosomal aberration. Fish are actually quite sensitive to the contaminants entering in their environment and it responds quite quickly to any kind of little contaminants. External morphology and internal anatomy of fish is adversely affected by the toxic substances. In the present study, Ctenopharyngodon idella was collected from Fisheries Research and Training institute Manawan, Lahore in order to study genotoxic effects of di methyl phthalate on fish health. Significant morphological abnormalities were not observed in fish. No scale loss, fin deformity and excessive mucous were observed in the fish. No fish died during the acclimation period before phthalate exposure, and no control fish died during toxicity tests. In the present study in vivo cytoxicity and genotoxicity assessment was performed using micronucleus in the peripheral erythrocytes of the fish, Ctenopharyngodon idella. As blood is the main index to analyze the toxicity level in the fish’s body. Hematological parameters were also analyzed to detect the level of toxicity in blood and to detect the immunological response generated by the fish as a reaction to the toxicant. Length weight relationship of the fish is used to calculate the growth parameters of the fish while condition factor is used to verify the health condition of fish in their habitat. Length weight values as well as condition factor were evaluated to determine the growth pattern and health status of fish status of the fish that was injected with phthalates. Fish growth parameters were analyzed by comparing them with the fish of the control group i.e. group which was not injected or provided with any kind of contamination. Maximum weight showed by the fish in control group was 39g and maximum length shown by the fish was 15.8cm respectively. While in treated group maximum weight shown by fish was 30g and maximum length shown by fish was 15.5cm respectively. Condition factor for the length weight parameters was also calculated. Condition factor basically shows the overall health status of the fish. Condition factor for control group was ranged from 0.65gcm-3 to 1.44gcm-3 and condition factor for treated group was ranged from 0.81gcm-3 to 0.97gcm-3 respectively. Growth parameters were studied by many scientists like Zantolli et al., 2009, they evaluated that the Fulton’s condition factor after exposing the fish Poecilia reticulate to Bis (2‐ethylhexyl) phthalate (DEHP) at different concentration and it remains unaffected. Results of these findings were contrary to our findings. Moreover Adeogun et al., 2018 supplied the fish Clarias gariepinus (African sharptooth catfish) to di-(2-ethylhexyl) phthalate (DEHP) and observed its effects for multiple days on the growth factor of the fish. In this study condition factor for both body weight and body length has decreased. Hence the findings of these studies are significantly similar to our findings because after exposure to di methyl phthalate for 6 days the condition factor for both body weight and body length has decreased in the treated group. Webster et al., 2010 studied the growth parameters and their study is significantly contradictory to our findings because the condition factor of the male zebra fish which was exposed to di-(2-ethylhexyl) phthalate (DEHP) for the period of 5 days did not change significantly but in our findings condition factor of growth parameter has significantly increased in treated group as compared to the control group. The haematological indices which are measured in the present study are a part of complete blood count (CBC). RBC’s, Hb and HCT are often used in the general evaluation of animal’s health. Changes in these indices from reference give an indication of disease. Hematological analysis was performed on the blood obtained from the fish that was injected with di-methyl-phthalate for 6 days. Different hematological indices like RBC’s, HB, HCT, PLT, MCV, MCH, and MCH were observed. Hemocytometer was used to count the peripheral erythrocytes. Hematological indices of both groups i.e. treated and control was analyzed and compared in order to determine the difference in values of these parameters. Total mean RBC’s count for treated group was 0.07±0.028 and for control group was 0.04. RBC’s total count for treated group was greater than the control group because of the stress response generated in fish of the treated group due to the injection of the toxicant. Similarly mean HB for treated and control group was 4.0 and 2.0 respectively.
Treated group had significantly higher HB value than the control group. Total mean value of hematocrit for treated group was 0.3±0.14 while total hematocrit value for control group was 0.2% which is also lower than the treated group. PLT’s total count for treated group was 1227.5±225.6 which is higher than the value of control group i.e.848. Total mean corpuscular volume for treated group was 42±2.82 while for control group it was 50.0. MCV of the treated group was lower than the control group. Total mean corpuscular hemoglobin for treated group was 6.2±2.54 and for control group it was 5.25. Treated group has high MCH value than the control group. Total value for the mean corpuscular hemoglobin concentration in treated and control group was 1500±707.1 and 1050 respectively. Value of MCHC in the blood of treated group was significantly higher than the blood of the fish from control group. Hematological parameters were studied by many scientists after exposing the fish to different kind of phthalates. Poopal et al., 2017 assessed the toxic effect of dibutyl phthalate (DBP) and diethyl phthalate (DEP) on the freshwater fish, Cyprinus carpio at different concentrations and these results were significantly different from our present study. The hematological parameters, such as hemoglobin (Hb), hematocrit (Hct) and erythrocyte (RBC), were found to decrease in the DBP and DEP treated fish, whereas their leucocyte (WBC) count increased compared to that of the control groups. A biphasic response is noted in the erythrocyte indices, such as mean cellular volume (MCV), mean cellular hemoglobin (MCH) and mean cellular hemoglobin concentration (MCHC) throughout the study period. Umamaheswari and Senthilnathan, 2013 analyzed the changes in the hematological indices of fish, Oreochromis mossambicus (Tilapia) after exposure to phthalate) and these results are quite consistent with our present study. Significant increase in mean red blood cells (RBC), mean white blood cells (WBC), mean haemoglobin, mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH), mean corpuscular haemogliobin concentration (MCHC) was observed in the blood of DEP treated fish when compared to the untreated ones. Javed et al., 2016 studied the hematological parameters of the Channa punctatus after its exposure to the toxicant. Findings of these studies are significantly different from the findings of our study as the values of Hb, RBC’s and MCH has decreased drastically in their treated groups but in our findings, the haematological parameters, Hb, RBC’s and MCH has increased in treated group after exposure to the di-methyl-phthalate as compared to the fish of the control group. All these elevated hematological parameters indicated that the production of red blood cells and white blood cells increased to strengthen the immunological system and enable it to reduce the stress caused by the phthalate exposure.
Genotoxicity is usually determined by the comet assay and micronucleus assay. The micronucleus test (MN) in fish erythrocytes has increasingly been used to detect the genotoxic effects of environmental mutagens and its frequency is considered to reflect the genotoxic damage to cells, mainly the chromosomes. Micronuclei are the small part of chromosomes left during anaphase stage of cell division due to some abnormality during their migration to opposite poles. In present study undertaken, percentage (0.62) of micronuclei induction was observed in erythrocytes of the fish after its exposure to the toxicant. Besides, morphologically altered erythrocyte is taken as an index of cytotoxicity. The genotoxic impact induced by the di methyl phthalate in Ctenopharyngodon idella was evaluated by using Micronucleus (MN) assay. It is quite helpful in the detection of micronuclei formed from the chromosomal loss in the anaphase of cell division and break down of chromosome during the process of cell division. MN assay has been widely used in different fish species of aquatic ecosystem. In the present study, blood was taken from the caudal vein of fish and transferred to EDTA tubes to determine the genotoxic impact of phthalate on fish blood. Nuclear alterations including MN were observed in the peripheral erythrocytes of fish injected with toxicant. The most frequently occurring nuclear lesions were segmented nuclei and binucleated nuclei. While clubbed shaped nuclei were least prevalent nuclear lesions. Notched shaped nuclei and dumbbeled shaped nuclei had the same frequency of 0.3%. Total mean frequency of nuclear lesions that were observed in the peripheral erythrocytes of the fish blood was 7.62±3.66 and average percentage of nuclear lesions was 0.62±0.32.
Induction of MN was studied by many scientists as a result of genotoxicity. Analysis of micronuclei, nuclear buds, and bi-polynucleated was performed in gills of blue mussels exposed for 3 weeks to sublethal concentrations of dialklyl phthalate by Barsiene et al., 2006 and results of these findings were consistent with the results of our study. Fourteen specimens from each treatment and control group were used for the analysis. Results demonstrated a significant increase in micronuclei frequency after the treatment with diallyl phthalate. Induction of bi-nucleated and nuclear buds was found in mussels exposed to diallyl phthalate. Bolognesi et al., 2006 studied the micronuclei induction in the peripheral erythrocytes of the fish exposed to dialkyl phthalate and the results are quite similar to our findings after exposure as chromosomal damage was determined as micronuclei (MN) frequency in fish erythrocytes. Nuclear anomalies such as blebbed, notched and lobed nuclei were also recorded in the peripheral erythrocytes of the fish. Revathy et al., 2019 studied the cytotoxic and genotoxic effects of two selected phthalates, in vivo and in vitro using fish Oreochromis mossambicus and these findings were quite consistent with our findings of the result as the exposure to di-methyl-phthalate also induced the formation of nuclear abnormalities. In vivo toxicological assessment of the phthalates showed genotoxicity of these phthalates at various concentrations, which was evident by the formation of micronuclei and other nuclear abnormalities in erythrocytes of the fish. Study showed the formation of micronuclei, nucleoplasmic bridge and nuclear buds in the blood cells of the fish.
- CONCLUSION
Based on our experimental findings, it is concluded that the toxicant i.e. Di-methyl-phthalate has caused physiological, immunological and morphological deformities in the fish Ctenopharyngodon idella. After injecting fish with di-methyl phthalate for 6 days, it was noticed that phthalate exposure caused behavioural changes in the fish. Hematological indices like RBC’s, Hb content, HCT value, MCH values has increased in the treated fish after exposure as fish’s body has responded to the toxicant by increasing its tolerance and immunity towards the phthalate. The results of the present investigation revealed that under experimental conditions, blood parameters of Ctenopharyngodon idella were sensitive to Di-methyl-phthalate exposure. Di-Methyl phthalate has also induced micronuclei and different nuclear abnormalities in the erythrocytes of the fish. Induction of micronuclei and other nuclear lesions proved the cytotoxic and genotoxic properties of Di methyl phthalate. So, Di-methyl phthalate is highly toxic to fish. Hence, the presence of di-methyl phthalate in waterways could have adverse impact on the survival of the fish. Further studies are recommended to determine and analyze the impact of phthalates on gene expression and DNA damage by the PCR technique and Comet assay, respectively. Therefore, it is necessary to monitor the levels of phthalates in aquatic environments and implement proper mitigation measures to reduce the negative effects of phthalate on fish, aquatic biota and humans at the top of the food chain through bioaccumulation, biomagnification and consumption of phthalate contaminated fish and other aquatic food products. It will not only increase the water quality but also helps to lessen the contamination of phthalates in aquatic organisms. Proper legislation regarding the monitoring of phthalates and guidelines for the permissible limits should be formulated and implemented by the authorities.
REFERENCES
- Adeniyi, A. A., Okedeyi, O. O. and Yusuf, K. A. 2011. Flame ionization gas chromatographic determination of phthalate esters in water, surface sediments and fish species in the Ogun river catchments, Ketu, Lagos, Nigeria. Environmental monitoring and assessment, 172(1-4): 561-569.
- Ali, D., Nagpure, N. S., Kumar, S., Kumar, R. and Kushwaha, B. 2008. Genotoxicity assessment of acute exposure of chlorpyrifos to freshwater fish Channa punctatus (Bloch) using micronucleus assay and alkaline single-cell gel electrophoresis. Chemosphere, 71(10): 1823-1831.
- Ali, D., Nagpure, N. S., Kumar, S., Kumar, R., Kushwaha, B. and Lakra, W. S. 2009. Assessment of genotoxic and mutagenic effects of chlorpyrifos in freshwater fish Channa punctatus (Bloch) using micronucleus assay and alkaline single-cell gel electrophoresis. Food and Chemical Toxicology, 47(3): 650-656.
- American Public Health Association, American Water Works Association, Water Pollution Control Federation, and Water Environment Federation. 1915. Standard methods for the examination of water and wastewater. 2: American Public Health Association.
- Arcangeli, A., Campana, I., Angeletti, D., Atzori, F., Azzolin, M., Carosso, L. and Paraboschi, M. 2018. Amount, composition, and spatial distribution of floating macro litter along fixed trans-border transects in the Mediterranean basin. Marine pollution bulletin, 129(2): 545-554.
- Azzouz, A. and Ballesteros, E. 2014. Trace analysis of endocrine disrupting compounds in environmental water samples by use of solid-phase extraction and gas chromatography with mass spectrometry detection. Journal of chromatography, 1360: 248-257.
- Bang, D. Y., Lee, I. K. and Lee, B. M. 2011. Toxicological characterization of phthalic acid. Toxicology, 27: 191–203.
- Baršienė, J., Rybakovas, A., Lang, T., Andreikėnaitė, L. and Michailovas, A. 2013. Environmental genotoxicity and cytotoxicity levels in fish from the North Sea offshore region and Atlantic coastal waters. Marine pollution bulletin, 68(1-2): 106-116.
- Baršienė, J., Šyvokienė, J. and Bjornstad, A. 2006. Induction of micronuclei and other nuclear abnormalities in mussels exposed to bisphenol A, diallyl phthalate and tetrabromodiphenyl ether-47. Aquatic toxicology, 78: S105-S108.
- Benli, A. Ç. K., Erkmen, B. and Erkoç, F. 2016. Genotoxicity of sub-lethal di-n-butyl phthalate (DBP) in Nile tilapia (Oreochromis niloticus)/Genotoksičnost subletalne koncentracije di-n-butil ftalata (DBP-a) u nilskoj tilapiji (Oreochromis niloticus). Archives of Industrial Hygiene and Toxicology, 67(1): 25-30.
- Boberg, J., Metzdorff, S., Wortziger, R., Axelstad, M., Brokken, L., Vinggaard, A. M. and Nellemann, C. 2008. Impact of diisobutyl phthalate and other PPAR agonists on steroidogenesis and plasma insulin and leptin levels in fetal rats. Toxicology, 250(2-3): 75-81.
- Bolognesi, C., Perrone, E., Roggieri, P., Pampanin, D. M. and Sciutto, A. 2006. Assessment of micronuclei induction in peripheral erythrocytes of fish exposed to xenobiotics under controlled conditions. Aquatic toxicology, 78: S93-S98.
- Çelik, E. Ş., Kaya, H., Yilmaz, S., Akbulut, M. and Tulgar, A. 2013. Effects of zinc exposure on the accumulation, haematology and immunology of Mozambique tilapia, Oreochromis mossambicus. African Journal of Biotechnology, 12(7).
- Chai, C., Cheng, H., Ge, W., Ma, D. and Shi, Y. 2014. Phthalic acid esters in soils from vegetable greenhouses in Shandong Peninsula, East China. PLoS One, 9(4): e95701.
- Chatterjee, S. and Karlovsky, P. 2010. Removal of the endocrine disrupter butyl benzyl phthalate from the environment. Applied microbiology and biotechnology, 87(1): 61-73.
- Chen, M. L., Chen, J. S., Tang, C. L. and Mao, I. F. 2008. The internal exposure of Taiwanese to phthalate—an evidence of intensive use of plastic materials. Environment International, 34(1): 79-85.
- Cravedi, J. P. and Perdu-Durand, E. 2002. The phthalate diesters DEHP and DBP do not induce lauric acid hydroxylase activity in rainbow trout. Marine environmental research, 54(3-5): 787-791.
- Cudmore, B. M. N. E. and Mandrak, N. E. 2004. Biological synopsis of grass carp (Ctenopharyngodon idella). Canadian manuscript report of fisheries and Aquatic Sciences, 2705(7).
- Dong, X., Qiu, X., Meng, S., Xu, H., Wu, X. and Yang, M. 2018. Proteomic profile and toxicity pathway analysis in zebrafish embryos exposed to bisphenol A and di-n-butyl phthalate at environmentally relevant levels. Chemosphere, 193: 313-320.
- Dudgeon, D., Arthington, A. H., Gessner, M. O., Kawabata, Z. I., Knowler, D. J., Lévêque, C. and Sullivan, C. A. 2006. Freshwater biodiversity: importance, threats, status and conservation challenges. Biological reviews, 81(2): 163-182.
- Fakayode, S. O. 2005. Impact assessment of industrial effluent on water quality of the receiving Alaro River in Ibadan, Nigeria. African Journal of Environmental Assessment and Management, 10: 1-13.
- FAO. 2018. The State of World Fisheries and Aquaculture 2018‐Meeting the sustainable development goals.
- Fatoki, O. S., Bornman, M., Ravandhalala, L., Chimuka, L., Genthe, B. and Adeniyi, A. 2010. Phthalate ester plasticizers in freshwater systems of Venda, South Africa and potential health effects. Water Sa, 36(1).
- Fenech, M. and Crott, J. W. 2002. Micronuclei, nucleoplasmic bridges and nuclear buds induced in folic acid deficient human lymphocytes—evidence for breakage–fusion-bridge cycles in the cytokinesis-block micronucleus assay. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis, 504(1-2): 131-136.
- Fenech, M., Chang, W. P., Kirsch-Volders, M., Holland, N., Bonassi, S. and Zeiger, E. 2003. HUMN project: detailed description of the scoring criteria for the cytokinesis-block micronucleus assay using isolated human lymphocyte cultures. Mutation Research/Genetic Toxicology and Environmental Mutagenesis, 534(1-2): 65-75.
- Food and Agriculture Organization of the United Nations. 2018. The State of World Fisheries and Aquaculture 2018–Meeting the sustainable development goals. FAO.
- França, J. K. G., Ranzani-Paiva, M. J. T., Lombardi, J. V., Carvalho, S. D. and Seriani, R. 2007. Toxicidade crônica do cloreto de mercurio associado ao selenio por meio do estudo hematológico em tilápia Oreochromis niloticus. Bioikos, 21(1): 11-19.
- Frederiksen, H., Skakkebaek, N. E. and Andersson, A. M. 2007. Metabolism of phthalates in humans. Molecular nutrition & food research, 51(7): 899-911.
- Gardner, S. T., Wood, A. T., Lester, R., Onkst, P. E., Burnham, N., Perygin, D. H. and Rayburn, J. 2016. Assessing differences in toxicity and teratogenicity of three phthalates, Diethyl phthalate, Di-n-propyl phthalate, and Di-n-butyl phthalate, using Xenopus laevis embryos. Journal of Toxicology and Environmental Health, Part A, 79(2): 71-82.
- Giovanoulis, G., Bui, T., Xu, F., Papadopoulou, E., Padilla-Sanchez, J. A., Covaci, A. and de Wit, C. A. 2018. Multi-pathway human exposure assessment of phthalate esters and DINCH. Environment international, 112: 115-126.
- Heudorf, U., Mersch-Sundermann, V. and Angerer, J. 2007. Phthalates: toxicology and exposure. International journal of hygiene and environmental health, 210(5): 623-634.
- Huang, B., Li, D. and Yang, Y. 2016. Joint toxicity of two phthalates with waterborne copper to Daphnia magna and Photobacterium phosphoreum. Bulletin of environmental contamination and toxicology, 97(3): 380-386.
- Huang, P. C., Tien, C. J., Sun, Y. M., Hsieh, C. Y. and Lee, C. C. 2008. Occurrence of phthalates in sediment and biota: relationship to aquatic factors and the biota-sediment accumulation factor. Chemosphere, 73(4): 539-544.
- Hussain, B., Sultana, T., Sultana, S., Masoud, M. S., Ahmed, Z. and Mahboob, S. 2018. Fish eco-genotoxicology: Comet and micronucleus assay in fish erythrocytes as in situ biomarker of freshwater pollution. Saudi journal of biological sciences, 25(2): 393-398.
- Iarmarcovai, G., Ceppi, M., Botta, A., Orsiere, T. and Bonassi, S. 2008. Micronuclei frequency in peripheral blood lymphocytes of cancer patients: a meta-analysis. Mutation Research/Reviews in Mutation Research, 659(3): 274-283.
- Karn, S. K. and Harada, H. 2001. Surface water pollution in three urban territories of Nepal, India, and Bangladesh. Environmental Management, 28(4): 483-496.
- Kessler, W., Numtip, W., Grote, K., Csanády, G. A., Chahoud, I. and Filser, J. G. 2004. Blood burden of di (2-ethylhexyl) phthalate and its primary metabolite mono (2-ethylhexyl) phthalate in pregnant and nonpregnant rats and marmosets. Toxicology and applied pharmacology, 195(2): 142-153.
- Khalil, S. R., Elhakim, Y. A. and EL-Murr, A. E. 2016. Sublethal concentrations of di-n-butyl phthalate promote biochemical changes and DNA damage in juvenile Nile tilapia (Oreochromis niloticus). Japanese Journal of Veterinary Research, 64(1): 67-80.
- Kirsch-Volders, M., Plas, G., Elhajouji, A., Lukamowicz, M., Gonzalez, L., Loock, K. V. and Decordier, I. 2011. The in vitro MN assay in 2011: origin and fate, biological significance, protocols, high throughput methodologies and toxicological relevance. Archives of toxicology, 85(8): 873-899.
- Kotti, M. E., Vlessidis, A. G., Thanasoulias, N. C. and Evmiridis, N. P. 2005. Assessment of river water quality in Northwestern Greece. Water Resources Management, 19(1): 77-94.
- Lee, I. K., and Lee, B. M. 2011. Toxicological characterization of phthalic acid. Toxicological research, 27(4): 191-203.
- Li, F. M., Wu, M., Yao, Y., Zheng, X., Zhao, J., Wang, Z. Y. and Xing, B. S. 2015. Inhibitory effects and oxidative target site of dibutyl phthalate on Karenia brevis. Chemosphere, 132: 32-39.
- LiLi, X., Fang, E. F., Scheibye-Knudsen, M., Cui, H., Qiu, L., Li, J. and Guo, H. 2014. Di-(2-ethylhexyl) phthalate inhibits DNA replication leading to hyperparylation, SIRT1 attenuation, and mitochondrial dysfunction in the testis. Scientific reports, 4: 6434.
- Liang, D. W., Zhang, T., Fang, H. H. and He, J. 2008. Phthalates biodegradation in the environment. Applied microbiology and Biotechnology, 80(2): 183.
- Lin, L., Dong, L., Meng, X., Li, Q., Huang, Z., Li, C. and Crittenden, J. 2018. Distribution and sources of polycyclic aromatic hydrocarbons and phthalic acid esters in water and surface sediment from the Three Gorges Reservoir. Journal of Environmental Sciences, 69: 271-280.
- Lindberg, H. K., Wang, X., Järventaus, H., Falck, G. C. M., Norppa, H., and Fenech, M. 2007. Origin of nuclear buds and micronuclei in normal and folate-deprived human lymphocytes. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis, 617(1-2): 33-45.
- Lu, Y., Lin, M., and Aitken, R. J. 2017. Exposure of spermatozoa to dibutyl phthalate induces abnormal embryonic development in a marine invertebrate Galeolaria caespitosa (Polychaeta: Serpulidae). Aquatic Toxicology, 191: 189-200.
- Mankidy, R., Wiseman, S., Ma, H. and Giesy, J. P. 2013. Biological impact of phthalates. Toxicology letters, 217(1): 50-58.
- Mateuca, R., Lombaert, N., Aka, P. V., Decordier, I. and Kirsch-Volders, M. 2006. Chromosomal changes: induction, detection methods and applicability in human biomonitoring. Biochimie, 88(11): 1515-1531.
- Molino, C., Filippi, S., Stoppiello, G. A., Meschini, R. and Angeletti, D. 2019. In vitro evaluation of cytotoxic and genotoxic effects of Di (2-ethylhexyl)-phthalate (DEHP) on European sea bass (Dicentrarchus labrax) embryonic cell line. Toxicology in Vitro, 56: 118-125.
- Mortensen, G. K., Main, K. M., Andersson, A. M., Leffers, H. and Skakkebæk, N. E. 2005. Determination of phthalate monoesters in human milk, consumer milk, and infant formula by tandem mass spectrometry (LC–MS–MS). Analytical and bioanalytical chemistry, 382(4): 1084-1092.
- Net, S., Delmont, A., Sempéré, R., Paluselli, A., and Ouddane, B. 2015. Reliable quantification of phthalates in environmental matrices (air, water, sludge, sediment and soil): a review. Science of the Total Environment, 515: 162-180.
- Pekey, H., Karakaş, D. and Bakoglu, M. 2004. Source apportionment of trace metals in surface waters of a polluted stream using multivariate statistical analyses. Marine Pollution Bulletin, 49(9-10): 809-818.
- Poopal, R. K., Ramesh, M., Maruthappan, V. and Babu Rajendran, R. 2017. Potential effects of low molecular weight phthalate esters (C16H22O4 and C12H14O4) on the freshwater fish Cyprinus carpio. Toxicology research, 6(4): 505-520.
- Qu, R., Feng, M., Sun, P. and Wang, Z. 2015. A comparative study on antioxidant status combined with integrated biomarker response in Carassius auratus fish exposed to nine phthalates. Environmental toxicology, 30(10): 1125-1134.
- Roslev, P., Vorkamp, K., Aarup, J., Frederiksen, K. and Nielsen, P. H. 2007. Degradation of phthalate esters in an activated sludge wastewater treatment plant. Water research, 41(5): 969-976.
- Sepperumal, U., and Saminathan, S. 2013. Effect of diethylphthalate on the haematological parameters of the freshwater fish Oreochromis mossambicus(Tilapia). Eurpeon Journal of Zoology, 2(4): 55-59.
- Seriani, R., França, J. G., Lombardi, J. V., Brito, J. M. and Ranzani-Paiva, M. J. T. 2015. Hematological changes and cytogenotoxicity in the tilapia Oreochromis niloticus caused by sub-chronic exposures to mercury and selenium. Fish physiology and biochemistry, 41(1): 311-322.
- Seyoum, A. and Pradhan, A. 2019. Effect of phthalates on development, reproduction, fat metabolism and lifespan in Daphnia magna. Science of the Total Environment, 654, 969-977.
- Singh, S. and Li, S. S. L. 2011. Phthalates: toxicogenomics and inferred human diseases. Genomics, 97(3): 148-157.
- Summak, S., Aydemir, N. C., Vatan, O., Yılmaz, D., Zorlu, T. and Bilaloğlu, R. 2010. Evaluation of genotoxicity from Nilufer Stream (Bursa/Turkey) water using piscine micronucleus test. Food and Chemical Toxicology, 48(8-9): 2443-2447.
- Uren-Webster, T. M., Lewis, C., Filby, A. L., Paull, G. C. and Santos, E. M. 2010. Mechanisms of toxicity of di (2-ethylhexyl) phthalate on the reproductive health of male zebrafish. Aquatic toxicology, 99(3): 360-369.
- Valton, A. S., Serre-Dargnat, C., Blanchard, M., Alliot, F., Chevreuil, M. and Teil, M. J. 2014. Determination of phthalates and their by-products in tissues of roach (Rutilus rutilus) from the Orge river (France). Environmental Science and Pollution Research, 21(22): 12723-12730.
- Wan, H. T., Leung, P. Y., Zhao, Y. G., Wei, X., Wong, M. H. and Wong, C. K. 2013. Blood plasma concentrations of endocrine disrupting chemicals in Hong Kong populations. Journal of hazardous materials, 261: 763-769.
- Wang, F., Xia, X., and Sha, Y. 2008. Distribution of phthalic acid esters in Wuhan section of the Yangtze River, China. Journal of Hazardous Materials, 154(1-3): 317-324.
- Wang, Y., Wang, T., Ban, Y., Shen, C., Shen, Q., Chai, X. and Wei, J. 2018. Di-(2-ethylhexyl) phthalate exposure modulates antioxidant enzyme activity and gene expression in juvenile and adult Daphnia magna. Archives of environmental contamination and toxicology, 75(1): 145-156.
- Wittassek, M., Koch, H. M., Angerer, J., and Brüning, T. 2011. Assessing exposure to phthalates–the human biomonitoring approach. Molecular nutrition & food research, 55(1): 7-31.
Publication History
Submitted: October 08, 2023
Accepted: October 11, 2023
Published: December 11, 2023
Identification
D-0172
Citation
Sidra Arshad, Lubna Rasheed, Tasneem Kausar & Aqsa Mubeen (2023). Assessment of Cytogenotoxicity in Ctenopharyngodon Idella Injected With Endocrine Disruptors. Dinkum Journal of Natural & Scientific Innovations, 2(12):815-851.
Copyright
© 2023 DJNSI. All rights reserved


